Stem Cell Therapy for Retinal Degeneration: The Evidence to Date
Affiliation.
- 1 Stem Cells and Cancer Biology Research Group, Department of Biosciences and Bioengineering, Indian Institute of Technology Guwahati, Guwahati, Assam, 781039, India.
- PMID: 34349498
- PMCID: PMC8327474
- DOI: 10.2147/BTT.S290331
There is a rise in the number of people who have vision loss due to retinal diseases, and conventional therapies for treating retinal degeneration fail to repair and regenerate the damaged retina. Several studies in animal models and human trials have explored the use of stem cells to repair the retinal tissue to improve visual acuity. In addition to the treatment of age-related macular degeneration (AMD) and diabetic retinopathy (DR), stem cell therapies were used to treat genetic diseases such as retinitis pigmentosa (RP) and Stargardt's disease, characterized by gradual loss of photoreceptor cells in the retina. Transplantation of retinal pigment epithelial (RPE) cells derived from embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) have shown promising results in improving retinal function in various preclinical models of retinal degeneration and clinical studies without any severe side effects. Mesenchymal stem cells (MSCs) were utilized to treat optic neuropathy, RP, DR, and glaucoma with positive clinical outcomes. This review summarizes the preclinical and clinical evidence of stem cell therapy and current limitations in utilizing stem cells for retinal degeneration.
Keywords: embryonic stem cells; induced pluripotent stem cells; mesenchymal stem cells; retinal degeneration; retinal pigment epithelial cells; retinitis pigmentosa.
© 2021 Sharma and Jaganathan.

Publication types
- For Authors
- Editorial Board
- Journals Home
To View More...
Purchase this article with an account.

- Defining Neural Progenitors as Stem Cells
- Neural Stem Cells-Progenitors in the Mammalian Eye
- Therapeutic Uses of Neural Stem Cells-Progenitors
- Iqbal Ahmad From the Department of Ophthalmology, University of Nebraska Medical Center, Omaha, Nebraska.
- Full Article
This feature is available to authenticated users only.
Iqbal Ahmad; Stem Cells: New Opportunities to Treat Eye Diseases. Invest. Ophthalmol. Vis. Sci. 2001;42(12):2743-2748.
Download citation file:
- Ris (Zotero)
- Reference Manager
© ARVO (1962-2015); The Authors (2016-present)
- Get Permissions
- Supplements
Related Articles
From other journals, related topics.
- Nanotechnology and Regenerative Medicine
- Eye Anatomy and Disorders
This PDF is available to Subscribers Only
You must be signed into an individual account to use this feature.
Advertisement
The Potential of Stem Cells as Treatment for Ocular Surface Diseases
- Ocular Surface (A Galor, Section Editor)
- Published: 11 November 2022
- Volume 10 , pages 209–217, ( 2022 )
Cite this article

- Andres Serrano 1 ,
- Kwaku A. Osei 1 ,
- Marcela Huertas-Bello 1 &
- Alfonso L. Sabater ORCID: orcid.org/0000-0001-6383-2558 1
214 Accesses
Explore all metrics
Purpose of Review
In this article, we review recent studies that examine stem cells as a potential treatment for ocular surface diseases.
Recent Findings
Mesenchymal stem cells (MSCs) derived from non-ocular surface tissues were effective in treating limbal stem cell deficiency and corneal endothelial dysfunction. In dry eye, limbal stem cells reduced ocular symptoms, while MSCs improved tear secretion and tear quality and reduced symptoms and inflammatory cytokine expression. There were no clinically significant adverse effects associated with stem cell treatment.
While available evidence supports stem cells as a potential treatment for ocular surface diseases, the applicability of these therapies in humans has yet to be fully established, given that over 80% of studies evaluating stem cell treatments have been carried out in animal models and non-human subjects. Future studies examining the safety and efficacy of stem cell therapies for ocular surface diseases in humans are thus warranted.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions
Similar content being viewed by others

Mesenchymal Stem Cells for Regeneration of the Ocular Surface

Pluripotent Stem Cells and Other Innovative Strategies for the Treatment of Ocular Surface Diseases
Mini review: human clinical studies of stem cell therapy in keratoconus
Papers of particular interest, published recently, have been highlighted as: • of importance •• of major importance.
The definition and classification of dry eye disease. report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):75–92.
Article Google Scholar
Narayanan S, Redfern RL, Miller WL, Nichols KK, McDermott AM. Dry eye disease and microbial keratitis: is there a connection? Ocul Surf. 2013;11(2):75–92.
Cavuoto KM, Stradiotto AC, Galor A. Role of the ocular surface microbiome in allergic disease. Curr Opin Allergy Clin Immunol. 2019;19(5):482–7.
Article CAS Google Scholar
Garza A, Diaz G, Hamdan M, Shetty A, Hong BY, Cervantes J. Homeostasis and defense at the surface of the eye. The conjunctival microbiota Curr Eye Res. 2021;46(1):1–6.
Li Z, Duan H, Jia Y, Zhao C, Li W, Wang X, et al. Long-term corneal recovery by simultaneous delivery of hPSC-derived corneal endothelial precursors and nicotinamide. J Clin Invest. 2022;132(1):e146658. This animal study shows that co-administered human embryonic stem cell-derived corneal endothelial precursors and nicotinamide are effective for corneal endothelial dysfunction .
Sun P, Shen L, Li YB, Du LQ, Wu XY. Long-term observation after transplantation of cultured human corneal endothelial cells for corneal endothelial dysfunction. Stem Cell Res Ther. 2022;13(1):228. This animal study shows that orbital adipose stem cell-derived hCECs have the capacity to regenerate a diseased corneal endothelium .
Wei LN, Wu CH, Lin CT, Liu IH. Topical applications of allogeneic adipose-derived mesenchymal stem cells ameliorate the canine keratoconjunctivitis sicca. BMC Vet Res. 2022;18(1):217. This animal study shows the therapeutic potential of adipose mesenchymal stem cells for dry eye treatment .
Shen K, Wang XJ, Liu KT, Li SH, Li J, Zhang JX, et al. Effects of exosomes from human adipose-derived mesenchymal stem cells on inflammatory response of mouse RAW264.7 cells and wound healing of full-thickness skin defects in mice. Zhonghua Shao Shang Za Zhi. 2022;38(3):215–26.
CAS Google Scholar
Wang G, Li H, Long H, Gong X, Hu S, Gong C. Exosomes derived from mouse adipose-derived mesenchymal stem cells alleviate benzalkonium chloride-induced mouse dry eye model via inhibiting NLRP3 inflammasome. Ophthalmic Res. 2022;65(1):40–51. This animal study shows that exoxomes from adipose stem cells have therapeutic potential in dry eye treatment .
Fu YS, Chen PR, Yeh CC, Pan JY, Kuo WC, Tseng KW. Human umbilical mesenchymal stem cell xenografts repair UV-induced photokeratitis in a rat model. Biomedicines. 2022;10(5):1125. This animal study shows the potential of umbilical cord-mesenchymal stem cells in treating photokeratitis .
Elhusseiny AM, Soleimani M, Eleiwa TK, ElSheikh RH, Frank CR, Naderan M, et al. Current and emerging therapies for limbal stem cell deficiency. Stem Cells Transl Med. 2022;11(3):259–68.
Kumar A, Yun H, Funderburgh ML, Du Y. Regenerative therapy for the cornea. Prog Retin Eye Res. 2022;87: 101011.
Ali M, Khan SY, Gottsch JD, Hutchinson EK, Khan A, Riazuddin SA. Pluripotent stem cell-derived corneal endothelial cells as an alternative to donor corneal endothelium in keratoplasty. Stem Cell Rep. 2021;16(9):2320–35. This animal shows the potential of cryopreserved human embryonic stem cell-derived endothelial cells to regenerate a dysfunctional corneal endothelium .
Hermida-Prieto M, Garcia-Castro J, Marinas-Pardo L. Systemic treatment of immune-mediated keratoconjunctivitis sicca with allogeneic stem cells improves the Schirmer tear test score in a canine spontaneous model of disease. J Clin Med. 2021;10(24):5981. This animal study shows that mesenchymal stem cells from adipose tissue improve tear secretion .
Moller-Hansen M, Larsen AC, Toft PB, Lynggaard CD, Schwartz C, Bruunsgaard H, et al. Safety and feasibility of mesenchymal stem cell therapy in patients with aqueous deficient dry eye disease. Ocul Surf. 2021;19:43–52. This clinical trial shows that allogeneic adipose mesenchymal stems can be an effective dry eye treatment .
Ruan Y, Jiang S, Musayeva A, Pfeiffer N, Gericke A. Corneal epithelial stem cells-physiology, pathophysiology and therapeutic options. Cells. 2021;10(9):2302.
Singh V, Tiwari A, Kethiri AR, Sangwan VS. Current perspectives of limbal-derived stem cells and its application in ocular surface regeneration and limbal stem cell transplantation. Stem Cells Transl Med. 2021;10(8):1121–8.
Rush SW, Chain J, Das H. Corneal epithelial stem cell supernatant in the treatment of severe dry eye disease: a pilot study. Clin Ophthalmol. 2021;15:3097–107. This clinical study shows the potential of donor limbal stem cells in treating limbal stem cell deficiency .
Falcao MSA, Brunel H, Peixer MAS, Dallago BSL, Costa FF, Queiroz LM, et al. Effect of allogeneic mesenchymal stem cells (MSCs) on corneal wound healing in dogs. J Tradit Complement Med. 2020;10(5):440–5. This animal study shows adipose tissue-derived mesenchymal stem cells as a treatment option for refractory corneal ulcer .
Ghareeb AE, Lako M, Figueiredo FC. Recent advances in stem cell therapy for limbal stem cell deficiency: a narrative review. Ophthalmol Ther. 2020;9(4):809–31.
He J, Ou S, Ren J, Sun H, He X, Zhao Z, et al. Tissue engineered corneal epithelium derived from clinical-grade human embryonic stem cells. Ocul Surf. 2020;18(4):672–80.
Calonge M, Perez I, Galindo S, Nieto-Miguel T, Lopez-Paniagua M, Fernandez I, et al. A proof-of-concept clinical trial using mesenchymal stem cells for the treatment of corneal epithelial stem cell deficiency. Transl Res. 2019;206:18–40.
Shukla S, Mittal SK, Foulsham W, Elbasiony E, Singhania D, Sahu SK, et al. Therapeutic efficacy of different routes of mesenchymal stem cell administration in corneal injury. Ocul Surf. 2019;17(4):729–36.
Galindo S, Herreras JM, Lopez-Paniagua M, Rey E, de la Mata A, Plata-Cordero M, et al. Therapeutic effect of human adipose tissue-derived mesenchymal stem cells in experimental corneal failure due to limbal stem cell niche damage. Stem Cells. 2017;35(10):2160–74.
Zhang C, Du L, Sun P, Shen L, Zhu J, Pang K, et al. Construction of tissue-engineered full-thickness cornea substitute using limbal epithelial cell-like and corneal endothelial cell-like cells derived from human embryonic stem cells. Biomaterials. 2017;124:180–94.
Bittencourt MK, Barros MA, Martins JF, Vasconcellos JP, Morais BP, Pompeia C, et al. Allogeneic mesenchymal stem cell transplantation in dogs with keratoconjunctivitis sicca. Cell Med. 2016;8(3):63–77.
Villatoro AJ, Fernandez V, Claros S, Rico-Llanos GA, Becerra J, Andrades JA. Use of adipose-derived mesenchymal stem cells in keratoconjunctivitis sicca in a canine model. Biomed Res Int. 2015;2015: 527926.
Ramos T, Scott D, Ahmad S. An update on ocular surface epithelial stem cells: cornea and conjunctiva. Stem Cells Int. 2015;2015: 601731.
Beyazyildiz E, Pinarli FA, Beyazyildiz O, Hekimoglu ER, Acar U, Demir MN, et al. Efficacy of topical mesenchymal stem cell therapy in the treatment of experimental dry eye syndrome model. Stem Cells Int. 2014;2014: 250230. This animal study shows the efficacy of mesenchymal stem cells in treating benzalkonium cholride-induced dry eye .
Weng J, He C, Lai P, Luo C, Guo R, Wu S, et al. Mesenchymal stromal cells treatment attenuates dry eye in patients with chronic graft-versus-host disease. Mol Ther. 2012;20(12):2347–54.
Damdimopoulou P, Rodin S, Stenfelt S, Antonsson L, Tryggvason K, Hovatta O. Human embryonic stem cells. Best Pract Res Clin Obstet Gynaecol. 2016;31:2–12.
Dhamodaran K, Subramani M, Ponnalagu M, Shetty R, Das D. Ocular stem cells: a status update! Stem Cell Res Ther. 2014;5(2):56.
Alison MR, Poulsom R, Forbes S, Wright NA. An introduction to stem cells. J Pathol. 2002;197(4):419–23.
Collin J, Queen R, Zerti D, Bojic S, Dorgau B, Moyse N, et al. A single cell atlas of human cornea that defines its development, limbal progenitor cells and their interactions with the immune cells. Ocul Surf. 2021;21:279–98.
Zakrzewski W, Dobrzynski M, Szymonowicz M, Rybak Z. Stem cells: past, present, and future. Stem Cell Res Ther. 2019;10(1):68.
Henningson CT Jr, Stanislaus MA, Gewirtz AM. 28 Embryonic and adult stem cell therapy. J Allergy Clin Immunol. 2003;111(2 Suppl):S745-53.
Bacakova L, Zarubova J, Travnickova M, Musilkova J, Pajorova J, Slepicka P, et al. Stem cells: their source, potency and use in regenerative therapies with focus on adipose-derived stem cells - a review. Biotechnol Adv. 2018;36(4):1111–26.
Kolios G, Moodley Y. Introduction to stem cells and regenerative medicine. Respiration. 2013;85(1):3–10.
Bremond-Gignac D, Copin H, Benkhalifa M. Corneal epithelial stem cells for corneal injury. Expert Opin Biol Ther. 2018;18(9):997–1003.
Marion NW, Mao JJ. Mesenchymal stem cells and tissue engineering. Methods Enzymol. 2006;420:339–61.
Pinnamaneni N, Funderburgh JL. Concise review: stem cells in the corneal stroma. Stem Cells. 2012;30(6):1059–63.
Levis HJ, Daniels JT. Recreating the human limbal epithelial stem cell niche with bioengineered limbal crypts. Curr Eye Res. 2016;41(9):1153–60.
Yoon JJ, Ismail S, Sherwin T. Limbal stem cells: central concepts of corneal epithelial homeostasis. World J Stem Cells. 2014;6(4):391–403.
Stewart RM, Sheridan CM, Hiscott PS, Czanner G, Kaye SB. Human conjunctival stem cells are predominantly located in the medial canthal and inferior forniceal areas. Invest Ophthalmol Vis Sci. 2015;56(3):2021–30.
Du Y, Funderburgh ML, Mann MM, SundarRaj N, Funderburgh JL. Multipotent stem cells in human corneal stroma. Stem Cells. 2005;23(9):1266–75.
Amano S, Yamagami S, Mimura T, Uchida S, Yokoo S. Corneal stromal and endothelial cell precursors. Cornea. 2006;25(10 Suppl 1):S73–7.
Yu WY, Sheridan C, Grierson I, Mason S, Kearns V, Lo AC, et al. Progenitors for the corneal endothelium and trabecular meshwork: a potential source for personalized stem cell therapy in corneal endothelial diseases and glaucoma. J Biomed Biotechnol. 2011;2011: 412743.
Samoila O, Samoila L. Stem cells in the path of light, from corneal to retinal reconstruction. Biomedicines. 2021;9(8):873.
Alviano F, Fossati V, Marchionni C, Arpinati M, Bonsi L, Franchina M, et al. Term amniotic membrane is a high throughput source for multipotent mesenchymal stem cells with the ability to differentiate into endothelial cells in vitro. BMC Dev Biol. 2007;7:11.
Paunescu V, Deak E, Herman D, Siska IR, Tanasie G, Bunu C, et al. In vitro differentiation of human mesenchymal stem cells to epithelial lineage. J Cell Mol Med. 2007;11(3):502–8.
Saghizadeh M, Kramerov AA, Svendsen CN, Ljubimov AV. Concise review: stem cells for corneal wound healing. Stem Cells. 2017;35(10):2105–14.
Uccelli A, Moretta L, Pistoia V. Immunoregulatory function of mesenchymal stem cells. Eur J Immunol. 2006;36(10):2566–73.
Bandeira F, Goh TW, Setiawan M, Yam GH, Mehta JS. Cellular therapy of corneal epithelial defect by adipose mesenchymal stem cell-derived epithelial progenitors. Stem Cell Res Ther. 2020;11(1):14.
Kuroda Y, Kitada M, Wakao S, Dezawa M. Bone marrow mesenchymal cells: how do they contribute to tissue repair and are they really stem cells? Arch Immunol Ther Exp (Warsz). 2011;59(5):369–78.
Ullah I, Subbarao RB, Rho GJ. Human mesenchymal stem cells - current trends and future prospective. Biosci Rep. 2015;35(2):e00191.
Call M, Meyer EA, Kao WW, Kruse FE, Schltzer-Schrehardt U. Murine hair follicle derived stem cell transplantation onto the cornea using a fibrin carrier. Bio Protoc. 2018;8(10): e2849.
Medvedev SP, Shevchenko AI, Zakian SM. Induced pluripotent stem cells: problems and advantages when applying them in regenerative medicine. Acta Naturae. 2010;2(2):18–28.
Gorecka J, Kostiuk V, Fereydooni A, Gonzalez L, Luo J, Dash B, et al. The potential and limitations of induced pluripotent stem cells to achieve wound healing. Stem Cell Res Ther. 2019;10(1):87.
Moradi S, Mahdizadeh H, Saric T, Kim J, Harati J, Shahsavarani H, et al. Research and therapy with induced pluripotent stem cells (iPSCs): social, legal, and ethical considerations. Stem Cell Res Ther. 2019;10(1):341.
Hayashi R, Ishikawa Y, Sasamoto Y, Katori R, Nomura N, Ichikawa T, et al. Co-ordinated ocular development from human iPS cells and recovery of corneal function. Nature. 2016;531(7594):376–80.
Hayashi R, Ishikawa Y, Katori R, Sasamoto Y, Taniwaki Y, Takayanagi H, et al. Coordinated generation of multiple ocular-like cell lineages and fabrication of functional corneal epithelial cell sheets from human iPS cells. Nat Protoc. 2017;12(4):683–96.
Thoft RA. Conjunctival transplantation. Arch Ophthalmol. 1977;95(8):1425–7.
Shanbhag SS, Patel CN, Goyal R, Donthineni PR, Singh V, Basu S. Simple limbal epithelial transplantation (SLET): review of indications, surgical technique, mechanism, outcomes, limitations, and impact. Indian J Ophthalmol. 2019;67(8):1265–77.
Soleimani M, Naderan M. Management strategies of ocular chemical burns: current perspectives. Clin Ophthalmol. 2020;14:2687–99.
Cullen AP. Photokeratitis and other phototoxic effects on the cornea and conjunctiva. Int J Toxicol. 2002;21(6):455–64.
Bergmanson JP. Corneal damage in photokeratitis–why is it so painful? Optom Vis Sci. 1990;67(6):407–13.
Wongvisavavit R, Parekh M, Ahmad S, Daniels JT. Challenges in corneal endothelial cell culture. Regen Med. 2021;16(9):871–91.
Eye Bank Association of America. 2020 Eye Banking Statistical Report. Washington, DC: Eye Bank Association of America; 2021.
Google Scholar
Ali M, Khan SY, Vasanth S, Ahmed MR, Chen R, Na CH, et al. Generation and proteome profiling of PBMC-originated, iPSC-derived corneal endothelial cells. Invest Ophthalmol Vis Sci. 2018;59(6):2437–44. This animal study shows that induced pluripotent stem cell-derived corneal endothelial cells (CECs) closely mimic human CECs .
Craig JP, Nichols KK, Akpek EK, Caffery B, Dua HS, Joo CK, et al. TFOS DEWS II definition and classification report. Ocul Surf. 2017;15(3):276–83.
Bron AJ, de Paiva CS, Chauhan SK, Bonini S, Gabison EE, Jain S, et al. TFOS DEWS II pathophysiology report. Ocul Surf. 2017;15(3):438–510.
Messmer EM, von Lindenfels V, Garbe A, Kampik A. Matrix metalloproteinase 9 testing in dry eye disease using a commercially available point-of-care immunoassay. Ophthalmology. 2016;123(11):2300–8.
Yamaguchi T. Inflammatory response in dry eye. Invest Ophthalmol Vis Sci. 2018;59(14):DES192–9.
Yoshida A, Fujihara T, Nakata K. Cyclosporin A increases tear fluid secretion via release of sensory neurotransmitters and muscarinic pathway in mice. Exp Eye Res. 1999;68(5):541–6.
Ames P, Galor A. Cyclosporine ophthalmic emulsions for the treatment of dry eye: a review of the clinical evidence. Clin Investig (Lond). 2015;5(3):267–85.
Song N, Scholtemeijer M, Shah K. Mesenchymal stem cell immunomodulation: mechanisms and therapeutic potential. Trends Pharmacol Sci. 2020;41(9):653–64.
Ren K. Exosomes in perspective: a potential surrogate for stem cell therapy. Odontology. 2019;107(3):271–84.
Li N, Zhao L, Wei Y, Ea VL, Nian H, Wei R. Recent advances of exosomes in immune-mediated eye diseases. Stem Cell Res Ther. 2019;10(1):278.
Zheng Q, Zhang S, Guo WZ, Li XK. The unique immunomodulatory properties of MSC-derived exosomes in organ transplantation. Front Immunol. 2021;12: 659621.
Chen W, Huang Y, Han J, Yu L, Li Y, Lu Z, et al. Immunomodulatory effects of mesenchymal stromal cells-derived exosome. Immunol Res. 2016;64(4):831–40.
Zheng Q, Ren Y, Reinach PS, She Y, Xiao B, Hua S, et al. Reactive oxygen species activated NLRP3 inflammasomes prime environment-induced murine dry eye. Exp Eye Res. 2014;125:1–8.
Zheng Q, Ren Y, Reinach PS, Xiao B, Lu H, Zhu Y, et al. Reactive oxygen species activated NLRP3 inflammasomes initiate inflammation in hyperosmolarity stressed human corneal epithelial cells and environment-induced dry eye patients. Exp Eye Res. 2015;134:133–40.
Zhou T, He C, Lai P, Yang Z, Liu Y, Xu H, et al. miR-204-containing exosomes ameliorate GVHD-associated dry eye disease. Sci Adv. 2022;8(2):eabj9617. This clinical trial shows that mesenchymal stem cell exoxomes can be effective in treating graft-versus-host disease dry eye .
Download references
Author information
Authors and affiliations.
Bascom Palmer Eye Institute, University of Miami Miller School of Medicine, 900 NW 17Th St., Miami, FL, 33136, USA
Andres Serrano, Kwaku A. Osei, Marcela Huertas-Bello & Alfonso L. Sabater
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Alfonso L. Sabater .
Ethics declarations
Conflict of interest.
The authors declare no competing interests.
Human/Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Ocular Surface
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Serrano, A., Osei, K.A., Huertas-Bello, M. et al. The Potential of Stem Cells as Treatment for Ocular Surface Diseases. Curr Ophthalmol Rep 10 , 209–217 (2022). https://doi.org/10.1007/s40135-022-00303-6
Download citation
Accepted : 24 October 2022
Published : 11 November 2022
Issue Date : December 2022
DOI : https://doi.org/10.1007/s40135-022-00303-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Corneal endothelial dysfunction
- Limbal stem cell deficiency
- Ocular surface disease
- Find a journal
- Publish with us
- Track your research
- U.S. Department of Health & Human Services

- Virtual Tour
- Staff Directory
- En Español
You are here
News releases.
News Release
Tuesday, January 2, 2018
NIH discovery brings stem cell therapy for eye disease closer to the clinic
Stem cell-derived retinal cells need primary cilia to support survival of light-sensing photoreceptors.

Scientists at the National Eye Institute (NEI), part of the National Institutes of Health, report that tiny tube-like protrusions called primary cilia on cells of the retinal pigment epithelium (RPE) — a layer of cells in the back of the eye — are essential for the survival of the retina’s light-sensing photoreceptors. The discovery has advanced efforts to make stem cell-derived RPE for transplantation into patients with geographic atrophy, otherwise known as dry age-related macular degeneration (AMD), a leading cause of blindness in the U.S. The study appears in the January 2 Cell Reports.
“We now have a better idea about how to generate and replace RPE cells, which appear to be among the first type of cells to stop working properly in AMD,” said the study’s lead investigator, Kapil Bharti, Ph.D., Stadtman investigator at the NEI. Bharti is leading the development of patient stem cell-derived RPE for an AMD clinical trial set to launch in 2018.
In a healthy eye, RPE cells nourish and support photoreceptors, the cells that convert light into electrical signals that travel to the brain via the optic nerve. RPE cells form a layer just behind the photoreceptors. In geographic atrophy, RPE cells die, which causes photoreceptors to degenerate, leading to vision loss.
Bharti and his colleagues are hoping to halt and reverse the progression of geographic atrophy by replacing diseased RPE with lab-made RPE. The approach involves using a patient’s blood cells to generate induced-pluripotent stem cells (iPSCs), cells capable of becoming any type of cell in the body. iPSCs are grown in the laboratory and then coaxed into becoming RPE for surgical implantation.
Attempts to create functional RPE implants, however, have hit a recurring obstacle: iPSCs programmed to become RPE cells have a tendency to get developmentally stuck, said Bharti. “The cells frequently fail to mature into functional RPE capable of supporting photoreceptors. In cases where they do mature, however, RPE maturation coincides with the emergence of primary cilia on the iPSC-RPE cells.”
The researchers tested three drugs known to modulate the growth of primary cilia on iPSC-derived RPE. As predicted, the two drugs known to enhance cilia growth significantly improved the structural and functional maturation of the iPSC-derived RPE. One important characteristic of maturity observed was that the RPE cells all oriented properly, correctly forming a single, functional monolayer. The iPSC-derived RPE cell gene expression profile also resembled that of adult RPE cells. And importantly, the cells performed a crucial function of mature RPE cells: they engulfed the tips of photoreceptor outer segments, a pruning process that keeps photoreceptors working properly.
By contrast, iPSC-derived RPE cells exposed to the third drug, an inhibitor of cilia growth, demonstrated severely disrupted structure and functionality.
As further confirmation of their observations, when the researchers genetically knocked down expression of cilia protein IFT88, the iPSC-derived RPE showed severe maturation and functional defects, as confirmed by gene expression analysis. Tissue staining showed that knocking down IFT88 led to reduced iPSC-derived RPE cell density and functional polarity, i.e., cells within the RPE tissue pointed in the wrong direction.
Bharti and his group found similar results in iPSC-derived lung cells, another type of epithelial cell with primary cilia. When iPSC-derived lung cells were exposed to drugs that enhance cilia growth, immunostaining confirmed that the cells looked structurally mature.
The report suggests that primary cilia regulate the suppression of the canonical WNT pathway, a cell signaling pathway involved in embryonic development. Suppression of the WNT pathway during RPE development instructs the cells to stop dividing and to begin differentiating into adult RPE, according to the researchers.
The researchers also generated iPSC-derived RPE from a patient with ciliopathy, a disorder that causes severe vision loss due to photoreceptor degeneration. The patient’s ciliopathy was associated with mutations of cilia gene CEP290. Compared to a healthy donor, iPSC-derived RPE from the ciliopathy patient had cilia that were smaller. The patient’s iPSC-derived RPE also had maturation and functional defects similar to those with IFT88 knockdown.
Further studies in a mouse model of ciliopathy confirmed an important temporal relationship: Looking across several early development stages, the RPE defects preceded the photoreceptor degeneration, which provides additional insights into ciliopathy-induced retinal degeneration.
The study’s findings have been incorporated into the group’s protocol for making clinical-grade iPSC-derived RPE. They will also inform the development of disease models for research of AMD and other degenerative retinal diseases, Bharti said.
This work was supported by the NEI intramural research program and the NIH Common Fund’s Regenerative Medicine Program.
NEI leads the federal government’s research on the visual system and eye diseases. NEI supports basic and clinical science programs to develop sight-saving treatments and address special needs of people with vision loss. For more information, visit https://www.nei.nih.gov .
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit www.nih.gov .
NIH…Turning Discovery Into Health ®
May-Simera, Helen Louise et al. “Primary Cilium Mediated Retinal Pigment Epithelium Maturation is Retarded in Ciliopathy Patient Cells”. Cell Reports. Published online January 2, 2018. DOI:10.1016/j.celrep.2017.12.038
Connect with Us
- More Social Media from NIH
Streamlining stem cells to treat macular degeneration
As we age, so do our eyes; most commonly, this involves changes to our vision and new glasses, but there are more severe forms of age-related eye problems. One of these is age-related macular degeneration, which affects the macula -- the back part of the eye that gives us sharp vision and the ability to distinguish details. The result is a blurriness in the central part of our visual field.
The macula is part of the eye's retina, which is the light-sensitive tissue mostly composed of the eye's visual cells: cone and rod photoreceptor cells. The retina also contains a layer called the retinal pigment epithelium (RPE), which has several important functions, including light absorption, cleaning up cellular waste, and keeping the other cells of the eye healthy.
The cells of the RPE also nourish and maintain the eye's photoreceptor cells, which is why one of the most promising treatment strategies for age-related macular degeneration is to replace aging, degenerating RPE cells with new ones grown from human embryonic stem cells.
Scientist have proposed several methods for converting stem cells into RPE, but there is still a gap in our knowledge of how cells respond to these stimuli over time. For example, some protocols take a few months while others can take up to a year. And yet, scientists are not clear as to what exactly happens over that period of time.
Mixed cell populations
"None of the differentiation protocols proposed for clinical trials have been scrutinized over time at the single-cell level -- we know they can make retinal pigment cells, but how cells evolve to that state remains a mystery," says Dr Gioele La Manno, a researcher with EPFL's Life Sciences Independent Research (ELISIR) program.
"Overall, the field has been so focused on the product of differentiation, that the path undertaken has been sometimes overlooked," he adds. "For the field to move forward, it is important to understand aspects of the dynamics of what happens in these protocols. The path to maturity could be as important as the end state, for example for the safety of treatment or for improving cell purity and reducing production time."
Tracking stem cells as they grow into RPE cells
La Manno has now led a study with Professor Fredrik Lanner at the Karolinska Institute (Sweden) profiling a protocol for differentiating human embryonic stem cells into RPE cells that is actually intended for clinical use. Their work shows that the protocol can develop safe and efficient pluripotent stem cell-based therapies for age-related macular degeneration. The study is published and featured on this month's cover of the journal Stem Cell Reports .
"Standard methods such as quantitative PCR and bulk RNA-seq capture the average expression of RNAs from large populations of cells," says Alex Lederer, a doctoral student at EPFL and one of the study's lead authors. "In mixed-cell populations, these measurements may obscure critical differences between individual cells that are important for knowing if the process is unfolding correctly." Instead, the researchers used a technique called single-cell RNA sequencing (scRNA-seq), which can detect all the active genes in an individual cell at a given time.
Looking at intermediate states
Using scRNA-seq, the researchers were able to study the entire gene expression profile of individual human embryonic stem cells throughout the differentiation protocol, which takes a total of sixty days. This allowed them to map out all the transient states within a population as they grew into retinal pigment cells, but also to optimize the protocol and suppress the growth of non-RPE cells, thus preventing the formation of contaminant cell populations. "The aim is to prevent mixed cell populations at the time of transplantation, and to make sure the cells at the endpoint are similar to original RPE cells from a patient's eye," says Lederer.
What they found was that on the way to becoming RPE cells, stem cells go through a process very similar to early embryonic development. During this, the cell culture took up a "rostral embryo patterning," the process that develops the embryo's neural tube, which will go on to become its brain and sensory systems for vision, hearing, and taste. After this patterning, the stem cells began to mature into RPE cells.
Eye-to-eye: transplanting RPE cells in an animal model
But the point of the differentiation protocol is to generate a pure population of RPE cells that can be implanted in patients' retinas to slow down macular degeneration. So the team transplanted their population of cells that had been monitored with scRNA-seq into the subretinal space of two female New Zealand white albino rabbits, which are what scientists in the field refer to as a "large-eyed animal model." The operation was carried out following approval by the Northern Stockholm Animal Experimental Ethics Committee.
The work showed that the protocol not only produces a pure RPE cell population but that those cells can continue maturing even after they have been transplanted in the subretinal space. "Our work shows that the differentiation protocol can develop safe and efficient pluripotent stem cell-based therapies for age-related macular degeneration," says Dr Fredrik Lanner, who is currently making sure the protocol can be soon used in clinics.
- Immune System
- Skin Cancer
- Hearing Impairment
- Schizophrenia
- Neuroscience
- Embryonic stem cell
- Adult stem cell
- Gene therapy
- Chemotherapy
- Stem cell treatments
- Bone marrow transplant
Story Source:
Materials provided by Ecole Polytechnique Fédérale de Lausanne . Original written by Nik Papageorgiou. Note: Content may be edited for style and length.
Journal Reference :
- Sandra Petrus-Reurer, Alex R. Lederer, Laura Baqué-Vidal, Iyadh Douagi, Belinda Pannagel, Irina Khven, Monica Aronsson, Hammurabi Bartuma, Magdalena Wagner, Andreas Wrona, Paschalis Efstathopoulos, Elham Jaberi, Hanni Willenbrock, Yutaka Shimizu, J. Carlos Villaescusa, Helder André, Erik Sundstrӧm, Aparna Bhaduri, Arnold Kriegstein, Anders Kvanta, Gioele La Manno, Fredrik Lanner. Molecular profiling of stem cell-derived retinal pigment epithelial cell differentiation established for clinical translation . Stem Cell Reports , 2022; 17 (6): 1458 DOI: 10.1016/j.stemcr.2022.05.005
Cite This Page :
Explore More
- Life Expectancy May Increase by 5 Years by 2050
- Toward a Successful Vaccine for HIV
- Highly Efficient Thermoelectric Materials
- Toward Human Brain Gene Therapy
- Whale Families Learn Each Other's Vocal Style
- AI Can Answer Complex Physics Questions
- Otters Use Tools to Survive a Changing World
- Monogamy in Mice: Newly Evolved Type of Cell
- Sustainable Electronics, Doped With Air
- Male Vs Female Brain Structure
Trending Topics
Strange & offbeat.
Taming Unruly Stem Cells to Enhance Eye Research
Widely Available Molecule Could Aid Development of Therapies for Blinding Diseases
By Brandon Levy
Tuesday, October 4, 2022
IRP researchers have discovered that a cheap, widely available molecule can help lab-grown stem cells develop into complex, three-dimensional models of the eye’s retina.
As scientists inch closer to growing fully functioning organs outside the body, it’s easy to forget that it’s already possible to grow miniature, simplified versions of some organs in the lab. These ‘organoids’ are an extremely useful research tool, but producing them can be tricky. New IRP research could make it much easier to grow organoids that mimic the eye’s retina, thereby accelerating discoveries about a variety of vision-impairing diseases. 1
Retinal organoids are three-dimensional structures with distinct layers containing all the major cell types found in the real human retina, the part of the eye that converts light into electrical signals the brain can interpret. While organoids aren’t perfect models, they are much closer to the real deal than the flat collections of a single cell type that scientists have long used for their experiments.
“A retinal organoid is as close to being a retina in a dish as we’ve ever had,” says IRP senior investigator Tiansen Li, Ph.D. , the new study’s senior author.
What’s more, scientists can grow retinal organoids that have the exact same genetic mutations as a patient with an eye disease, enabling them to investigate the biological causes of a person’s specific vision problems. This involves taking skin or blood cells from the patient and turning them into cells called induced pluripotent stem cells (iPSCs), which can develop into any cell type found in the body. These iPSCs can then be grown in unlimited quantities to create ‘cell lines’ with specific properties, and researchers like those in Dr. Li’s lab coax them to develop into retinal organoids by exposing them to specific molecules. Unfortunately, not all cell lines cooperate with the scientists trying to manipulate them.
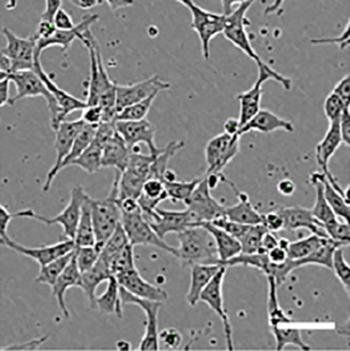
Human pluripotent stem cells, as seen under a microscope.
“The cell lines that we create from different people can react differently to our process,” says IRP postdoctoral fellow Florian Regent, Ph.D., who led the new study in Dr. Li’s lab. “Our process can be very effective for some cell lines and not for others. And, of course, when you want to study a particular patient’s cell line, you can’t work with a different one — the process has to work with that cell line.”
In the new study, Dr. Li’s team added an additional chemical to their standard process for turning iPSCs into retinal organoids: a form of vitamin B3 called nicotinamide that is commonly found in dietary supplements. The group selected nicotinamide for the experiment because it has previously been used to encourage iPSCs to develop into a different part of the retina, the retinal pigment epithelium (RPE).
At the beginning of their experiment, the IRP researchers exposed stem cells from eight different cell lines to nicotinamide for either eight or twenty one days. Some of the cell lines readily developed into retinal organoids even without the nicotinamide treatment, while others did not. The researchers discovered that the eight-day nicotinamide treatment significantly increased the yield of retinal organoids from all of the cell lines, including those that produced very few organoids in the absence of nicotinamide. The three-week treatment also boosted organoid yields from some of the cell lines, but decreased the yield for one cell line, suggesting that nicotinamide treatment is more beneficial during a specific, narrow developmental window.
“During development, cells are influenced by their intrinsic programs, obviously, but also by external environmental cues,” Dr. Li explains. “These cues act at defined time points, so a cue that does one thing at one point in time, two days later might do the opposite. That applies to nicotinamide as well. If you apply it for an extended duration, it might overstay its welcome.”
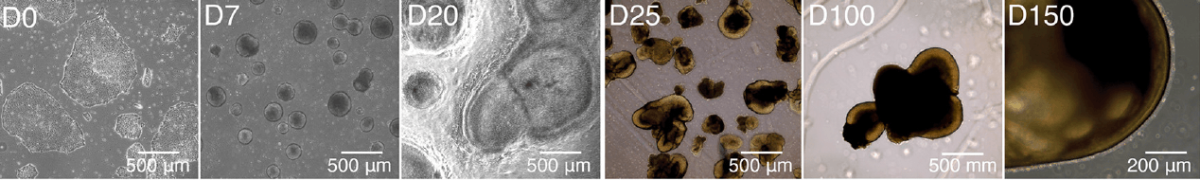
These images from the study show how induced pluripotent stem cells develop into retinal organoids over the course of 150 days.
The study also shed light on why nicotinamide encourages iPSCs to develop into retinal organoids. During the early stages of the experiment, the stem cells treated with nicotinamide more readily transformed into an immature form of neural cells capable of eventually becoming retinal cells, as opposed to changing into the sorts of immature cells that could only develop into cell types like skin and bone cells. Moreover, the researchers discovered that nicotinamide-treated cells had lower activity in a set of genes related to bone morphogenic protein (BMP), a molecule that plays an important role in embryonic development. This discovery suggests that inhibition of this BMP-related cellular system is one reason — though likely not the only reason — why nicotinamide helps turn stem cells into retinal organoids.

Dr. Florian Regent (left) and Dr. Tiansen Li (right)
Regardless of how exactly nicotinamide works its magic, Dr. Li believes that it could soon become a standard part of the procedures other labs use to produce retinal organoids. Nicotinamide is cheap and widely available, and by using it to facilitate the creation of retinal organoids from stem cell lines that previously refused to develop into them, scientists could find it much easier to make discoveries about a number of diseases that affect vision.
“We continue to gather data to support the validity of this approach, and our hope and expectation is that the entire field will adopt it, just as they adopted other standardized and widely used additions to retinal organoid production,” Dr. Li says. “I would like to see it used in everybody’s procedures in the next few years. That would make me very happy.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
References:
[1] Regent F, Batz Z, Kelley RA, Gieser L, Swaroop A, Chen HY, Li T. Nicotinamide Promotes Formation of Retinal Organoids From Human Pluripotent Stem Cells via Enhanced Neural Cell Fate Commitment. Front Cell Neurosci . 2022 Jun 17;16:878351. doi: 10.3389/fncel.2022.878351.
Related Blog Posts
- Experimental Treatment Helps Neurons Recover From Damage
- Pain Research Center Accelerates IRP Pain Studies
- Tiny Molecules Have Big Potential for Treating Eye Diseases
- Postdoc Profile: A Bird’s-Eye View of Retinal Disease
- Non-Toxic Drug Could Increase Availability of Organ Transplants
This page was last updated on Wednesday, May 24, 2023
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- RESEARCH HIGHLIGHT
- 09 May 2024
CRISPR therapy restores some vision to people with blindness
The light-absorbing rods and cones in the retina (artificially coloured) deteriorate in people with Leber’s congenital amaurosis. Credit: Science Photo Library
A CRISPR-based gene-editing therapy led to improved vision in people with an inherited condition that causes blindness 1 .
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
24,99 € / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
185,98 € per year
only 3,65 € per issue
Rent or buy this article
Prices vary by article type
Prices may be subject to local taxes which are calculated during checkout
Nature 629 , 507 (2024)
doi: https://doi.org/10.1038/d41586-024-01285-0
Pierce, E. A. et al. N. Engl. J. Med . https://doi.org/10.1056/NEJMoa2309915 (2024).
Article Google Scholar
Download references
- Gene therapy
How to kill the ‘zombie’ cells that make you age
News Feature 15 MAY 24

Targeting RNA opens therapeutic avenues for Timothy syndrome
News & Views 24 APR 24
Improving prime editing with an endogenous small RNA-binding protein
Article 03 APR 24
Faculty Positions& Postdoctoral Research Fellow, School of Optical and Electronic Information, HUST
Job Opportunities: Leading talents, young talents, overseas outstanding young scholars, postdoctoral researchers.
Wuhan, Hubei, China
School of Optical and Electronic Information, Huazhong University of Science and Technology

Postdoc in CRISPR Meta-Analytics and AI for Therapeutic Target Discovery and Priotisation (OT Grant)
APPLICATION CLOSING DATE: 14/06/2024 Human Technopole (HT) is a new interdisciplinary life science research institute created and supported by the...
Human Technopole
Research Associate - Metabolism
Houston, Texas (US)
Baylor College of Medicine (BCM)
Postdoc Fellowships
Train with world-renowned cancer researchers at NIH? Consider joining the Center for Cancer Research (CCR) at the National Cancer Institute
Bethesda, Maryland
NIH National Cancer Institute (NCI)
Faculty Recruitment, Westlake University School of Medicine
Faculty positions are open at four distinct ranks: Assistant Professor, Associate Professor, Full Professor, and Chair Professor.
Hangzhou, Zhejiang, China
Westlake University
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies

- For Ophthalmologists
- For Practice Management
- For Clinical Teams
- For Public & Patients
Museum of the Eye
- Eye Health A-Z
- Glasses & Contacts
- Tips & Prevention
- Ask an Ophthalmologist
- Patient Stories
- No-Cost Eye Exams
- For Public & Patients /

Stem Cell Therapy for Eye Disease: What You Need to Know
Avoid unlicensed clinics offering unapproved stem cell therapy.
Stem cell therapies are getting headlines for their potential to cure diseases, including those that affect vision. But an important message is missing: the therapies are not yet proven to be safe and effective for your eyes .
Stem cell treatments appear to offer hope to people with few options to recover vision. This includes people with forms of age-related macular degeneration (AMD) , retinitis pigmentosa (RP) , and Stargardt disease . Some clinics across the United States offer "stem-cell therapy" to people outside of clinical trials. But the Food and Drug Administration (FDA) has not approved the treatments they offer. These treatments often use unproven products that may be ineffective or dangerous. These products may carry serious risks, including tumor growth.
Ask These Questions If You’re Considering Stem Cell Therapy for Eye Disease
It is important that you know that there are no stem cell products approved by the FDA for eye disease right now. If you want stem cell therapy, look for a clinical trial and discuss the matter with your ophthalmologist. A clinic should not expect you to pay thousands of dollars for an unproven, unapproved therapy. Your health insurance will not cover the cost of an unapproved treatment.
Before agreeing to a stem cell treatment, ask yourself:
- Is the stem cell treatment approved by the FDA?
- Is the stem cell treatment part of an FDA-approved clinical trial?
- Is the stem cell treatment covered by your health insurance?
It is frustrating and frightening to face the loss of vision while waiting for potential treatments. However, choosing to pursue an unproven treatment in an unlicensed clinic is an unacceptable risk to your vision and your overall health.
The American Academy of Ophthalmology wants to reduce or eliminate unlicensed stem cell clinics in the United States. In June 2016, the Academy asked the FDA to tighten regulations and increase investigations into stem cell treatments given outside of clinical trials.
Stem Cells in the News
- Unregulated Stem Cell Treatments Can Be Dangerous
- Stem Cell Treatment for Dry AMD Moves Closer to Human Trials
- Stem Cells May Return Some Vision Lost to Wet AMD
- Could Stem Cells Cure Blindness Caused by Macular Degeneration?
- Will Stem Cell or Gene Therapies Bring Cures for Retinal Diseases?
- Find an Ophthalmologist Search Advanced Search
Free EyeSmart Newsletter
All content on the Academy’s website is protected by copyright law and the Terms of Service . This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
- About the Academy
- Jobs at the Academy
- Financial Relationships with Industry
- Medical Disclaimer
- Privacy Policy
- Terms of Service
- Statement on Artificial Intelligence
- For Advertisers
- Ophthalmology Job Center
FOLLOW THE ACADEMY
Medical Professionals
Public & Patients
- New Treatments for Age-Related Macular Degeneration
- New Treatments for Retinitis Pigmentosa

Cultural Relativity and Acceptance of Embryonic Stem Cell Research
Article sidebar.
Main Article Content
There is a debate about the ethical implications of using human embryos in stem cell research, which can be influenced by cultural, moral, and social values. This paper argues for an adaptable framework to accommodate diverse cultural and religious perspectives. By using an adaptive ethics model, research protections can reflect various populations and foster growth in stem cell research possibilities.
INTRODUCTION
Stem cell research combines biology, medicine, and technology, promising to alter health care and the understanding of human development. Yet, ethical contention exists because of individuals’ perceptions of using human embryos based on their various cultural, moral, and social values. While these disagreements concerning policy, use, and general acceptance have prompted the development of an international ethics policy, such a uniform approach can overlook the nuanced ethical landscapes between cultures. With diverse viewpoints in public health, a single global policy, especially one reflecting Western ethics or the ethics prevalent in high-income countries, is impractical. This paper argues for a culturally sensitive, adaptable framework for the use of embryonic stem cells. Stem cell policy should accommodate varying ethical viewpoints and promote an effective global dialogue. With an extension of an ethics model that can adapt to various cultures, we recommend localized guidelines that reflect the moral views of the people those guidelines serve.
Stem cells, characterized by their unique ability to differentiate into various cell types, enable the repair or replacement of damaged tissues. Two primary types of stem cells are somatic stem cells (adult stem cells) and embryonic stem cells. Adult stem cells exist in developed tissues and maintain the body’s repair processes. [1] Embryonic stem cells (ESC) are remarkably pluripotent or versatile, making them valuable in research. [2] However, the use of ESCs has sparked ethics debates. Considering the potential of embryonic stem cells, research guidelines are essential. The International Society for Stem Cell Research (ISSCR) provides international stem cell research guidelines. They call for “public conversations touching on the scientific significance as well as the societal and ethical issues raised by ESC research.” [3] The ISSCR also publishes updates about culturing human embryos 14 days post fertilization, suggesting local policies and regulations should continue to evolve as ESC research develops. [4] Like the ISSCR, which calls for local law and policy to adapt to developing stem cell research given cultural acceptance, this paper highlights the importance of local social factors such as religion and culture.
I. Global Cultural Perspective of Embryonic Stem Cells
Views on ESCs vary throughout the world. Some countries readily embrace stem cell research and therapies, while others have stricter regulations due to ethical concerns surrounding embryonic stem cells and when an embryo becomes entitled to moral consideration. The philosophical issue of when the “someone” begins to be a human after fertilization, in the morally relevant sense, [5] impacts when an embryo becomes not just worthy of protection but morally entitled to it. The process of creating embryonic stem cell lines involves the destruction of the embryos for research. [6] Consequently, global engagement in ESC research depends on social-cultural acceptability.
a. US and Rights-Based Cultures
In the United States, attitudes toward stem cell therapies are diverse. The ethics and social approaches, which value individualism, [7] trigger debates regarding the destruction of human embryos, creating a complex regulatory environment. For example, the 1996 Dickey-Wicker Amendment prohibited federal funding for the creation of embryos for research and the destruction of embryos for “more than allowed for research on fetuses in utero.” [8] Following suit, in 2001, the Bush Administration heavily restricted stem cell lines for research. However, the Stem Cell Research Enhancement Act of 2005 was proposed to help develop ESC research but was ultimately vetoed. [9] Under the Obama administration, in 2009, an executive order lifted restrictions allowing for more development in this field. [10] The flux of research capacity and funding parallels the different cultural perceptions of human dignity of the embryo and how it is socially presented within the country’s research culture. [11]
b. Ubuntu and Collective Cultures
African bioethics differs from Western individualism because of the different traditions and values. African traditions, as described by individuals from South Africa and supported by some studies in other African countries, including Ghana and Kenya, follow the African moral philosophies of Ubuntu or Botho and Ukama , which “advocates for a form of wholeness that comes through one’s relationship and connectedness with other people in the society,” [12] making autonomy a socially collective concept. In this context, for the community to act autonomously, individuals would come together to decide what is best for the collective. Thus, stem cell research would require examining the value of the research to society as a whole and the use of the embryos as a collective societal resource. If society views the source as part of the collective whole, and opposes using stem cells, compromising the cultural values to pursue research may cause social detachment and stunt research growth. [13] Based on local culture and moral philosophy, the permissibility of stem cell research depends on how embryo, stem cell, and cell line therapies relate to the community as a whole . Ubuntu is the expression of humanness, with the person’s identity drawn from the “’I am because we are’” value. [14] The decision in a collectivistic culture becomes one born of cultural context, and individual decisions give deference to others in the society.
Consent differs in cultures where thought and moral philosophy are based on a collective paradigm. So, applying Western bioethical concepts is unrealistic. For one, Africa is a diverse continent with many countries with different belief systems, access to health care, and reliance on traditional or Western medicines. Where traditional medicine is the primary treatment, the “’restrictive focus on biomedically-related bioethics’” [is] problematic in African contexts because it neglects bioethical issues raised by traditional systems.” [15] No single approach applies in all areas or contexts. Rather than evaluating the permissibility of ESC research according to Western concepts such as the four principles approach, different ethics approaches should prevail.
Another consideration is the socio-economic standing of countries. In parts of South Africa, researchers have not focused heavily on contributing to the stem cell discourse, either because it is not considered health care or a health science priority or because resources are unavailable. [16] Each country’s priorities differ given different social, political, and economic factors. In South Africa, for instance, areas such as maternal mortality, non-communicable diseases, telemedicine, and the strength of health systems need improvement and require more focus. [17] Stem cell research could benefit the population, but it also could divert resources from basic medical care. Researchers in South Africa adhere to the National Health Act and Medicines Control Act in South Africa and international guidelines; however, the Act is not strictly enforced, and there is no clear legislation for research conduct or ethical guidelines. [18]
Some parts of Africa condemn stem cell research. For example, 98.2 percent of the Tunisian population is Muslim. [19] Tunisia does not permit stem cell research because of moral conflict with a Fatwa. Religion heavily saturates the regulation and direction of research. [20] Stem cell use became permissible for reproductive purposes only recently, with tight restrictions preventing cells from being used in any research other than procedures concerning ART/IVF. Their use is conditioned on consent, and available only to married couples. [21] The community's receptiveness to stem cell research depends on including communitarian African ethics.
c. Asia
Some Asian countries also have a collective model of ethics and decision making. [22] In China, the ethics model promotes a sincere respect for life or human dignity, [23] based on protective medicine. This model, influenced by Traditional Chinese Medicine (TCM), [24] recognizes Qi as the vital energy delivered via the meridians of the body; it connects illness to body systems, the body’s entire constitution, and the universe for a holistic bond of nature, health, and quality of life. [25] Following a protective ethics model, and traditional customs of wholeness, investment in stem cell research is heavily desired for its applications in regenerative therapies, disease modeling, and protective medicines. In a survey of medical students and healthcare practitioners, 30.8 percent considered stem cell research morally unacceptable while 63.5 percent accepted medical research using human embryonic stem cells. Of these individuals, 89.9 percent supported increased funding for stem cell research. [26] The scientific community might not reflect the overall population. From 1997 to 2019, China spent a total of $576 million (USD) on stem cell research at 8,050 stem cell programs, increased published presence from 0.6 percent to 14.01 percent of total global stem cell publications as of 2014, and made significant strides in cell-based therapies for various medical conditions. [27] However, while China has made substantial investments in stem cell research and achieved notable progress in clinical applications, concerns linger regarding ethical oversight and transparency. [28] For example, the China Biosecurity Law, promoted by the National Health Commission and China Hospital Association, attempted to mitigate risks by introducing an institutional review board (IRB) in the regulatory bodies. 5800 IRBs registered with the Chinese Clinical Trial Registry since 2021. [29] However, issues still need to be addressed in implementing effective IRB review and approval procedures.
The substantial government funding and focus on scientific advancement have sometimes overshadowed considerations of regional cultures, ethnic minorities, and individual perspectives, particularly evident during the one-child policy era. As government policy adapts to promote public stability, such as the change from the one-child to the two-child policy, [30] research ethics should also adapt to ensure respect for the values of its represented peoples.
Japan is also relatively supportive of stem cell research and therapies. Japan has a more transparent regulatory framework, allowing for faster approval of regenerative medicine products, which has led to several advanced clinical trials and therapies. [31] South Korea is also actively engaged in stem cell research and has a history of breakthroughs in cloning and embryonic stem cells. [32] However, the field is controversial, and there are issues of scientific integrity. For example, the Korean FDA fast-tracked products for approval, [33] and in another instance, the oocyte source was unclear and possibly violated ethical standards. [34] Trust is important in research, as it builds collaborative foundations between colleagues, trial participant comfort, open-mindedness for complicated and sensitive discussions, and supports regulatory procedures for stakeholders. There is a need to respect the culture’s interest, engagement, and for research and clinical trials to be transparent and have ethical oversight to promote global research discourse and trust.
d. Middle East
Countries in the Middle East have varying degrees of acceptance of or restrictions to policies related to using embryonic stem cells due to cultural and religious influences. Saudi Arabia has made significant contributions to stem cell research, and conducts research based on international guidelines for ethical conduct and under strict adherence to guidelines in accordance with Islamic principles. Specifically, the Saudi government and people require ESC research to adhere to Sharia law. In addition to umbilical and placental stem cells, [35] Saudi Arabia permits the use of embryonic stem cells as long as they come from miscarriages, therapeutic abortions permissible by Sharia law, or are left over from in vitro fertilization and donated to research. [36] Laws and ethical guidelines for stem cell research allow the development of research institutions such as the King Abdullah International Medical Research Center, which has a cord blood bank and a stem cell registry with nearly 10,000 donors. [37] Such volume and acceptance are due to the ethical ‘permissibility’ of the donor sources, which do not conflict with religious pillars. However, some researchers err on the side of caution, choosing not to use embryos or fetal tissue as they feel it is unethical to do so. [38]
Jordan has a positive research ethics culture. [39] However, there is a significant issue of lack of trust in researchers, with 45.23 percent (38.66 percent agreeing and 6.57 percent strongly agreeing) of Jordanians holding a low level of trust in researchers, compared to 81.34 percent of Jordanians agreeing that they feel safe to participate in a research trial. [40] Safety testifies to the feeling of confidence that adequate measures are in place to protect participants from harm, whereas trust in researchers could represent the confidence in researchers to act in the participants’ best interests, adhere to ethical guidelines, provide accurate information, and respect participants’ rights and dignity. One method to improve trust would be to address communication issues relevant to ESC. Legislation surrounding stem cell research has adopted specific language, especially concerning clarification “between ‘stem cells’ and ‘embryonic stem cells’” in translation. [41] Furthermore, legislation “mandates the creation of a national committee… laying out specific regulations for stem-cell banking in accordance with international standards.” [42] This broad regulation opens the door for future global engagement and maintains transparency. However, these regulations may also constrain the influence of research direction, pace, and accessibility of research outcomes.
e. Europe
In the European Union (EU), ethics is also principle-based, but the principles of autonomy, dignity, integrity, and vulnerability are interconnected. [43] As such, the opportunity for cohesion and concessions between individuals’ thoughts and ideals allows for a more adaptable ethics model due to the flexible principles that relate to the human experience The EU has put forth a framework in its Convention for the Protection of Human Rights and Dignity of the Human Being allowing member states to take different approaches. Each European state applies these principles to its specific conventions, leading to or reflecting different acceptance levels of stem cell research. [44]
For example, in Germany, Lebenzusammenhang , or the coherence of life, references integrity in the unity of human culture. Namely, the personal sphere “should not be subject to external intervention.” [45] Stem cell interventions could affect this concept of bodily completeness, leading to heavy restrictions. Under the Grundgesetz, human dignity and the right to life with physical integrity are paramount. [46] The Embryo Protection Act of 1991 made producing cell lines illegal. Cell lines can be imported if approved by the Central Ethics Commission for Stem Cell Research only if they were derived before May 2007. [47] Stem cell research respects the integrity of life for the embryo with heavy specifications and intense oversight. This is vastly different in Finland, where the regulatory bodies find research more permissible in IVF excess, but only up to 14 days after fertilization. [48] Spain’s approach differs still, with a comprehensive regulatory framework. [49] Thus, research regulation can be culture-specific due to variations in applied principles. Diverse cultures call for various approaches to ethical permissibility. [50] Only an adaptive-deliberative model can address the cultural constructions of self and achieve positive, culturally sensitive stem cell research practices. [51]
II. Religious Perspectives on ESC
Embryonic stem cell sources are the main consideration within religious contexts. While individuals may not regard their own religious texts as authoritative or factual, religion can shape their foundations or perspectives.
The Qur'an states:
“And indeed We created man from a quintessence of clay. Then We placed within him a small quantity of nutfa (sperm to fertilize) in a safe place. Then We have fashioned the nutfa into an ‘alaqa (clinging clot or cell cluster), then We developed the ‘alaqa into mudgha (a lump of flesh), and We made mudgha into bones, and clothed the bones with flesh, then We brought it into being as a new creation. So Blessed is Allah, the Best of Creators.” [52]
Many scholars of Islam estimate the time of soul installment, marked by the angel breathing in the soul to bring the individual into creation, as 120 days from conception. [53] Personhood begins at this point, and the value of life would prohibit research or experimentation that could harm the individual. If the fetus is more than 120 days old, the time ensoulment is interpreted to occur according to Islamic law, abortion is no longer permissible. [54] There are a few opposing opinions about early embryos in Islamic traditions. According to some Islamic theologians, there is no ensoulment of the early embryo, which is the source of stem cells for ESC research. [55]
In Buddhism, the stance on stem cell research is not settled. The main tenets, the prohibition against harming or destroying others (ahimsa) and the pursuit of knowledge (prajña) and compassion (karuna), leave Buddhist scholars and communities divided. [56] Some scholars argue stem cell research is in accordance with the Buddhist tenet of seeking knowledge and ending human suffering. Others feel it violates the principle of not harming others. Finding the balance between these two points relies on the karmic burden of Buddhist morality. In trying to prevent ahimsa towards the embryo, Buddhist scholars suggest that to comply with Buddhist tenets, research cannot be done as the embryo has personhood at the moment of conception and would reincarnate immediately, harming the individual's ability to build their karmic burden. [57] On the other hand, the Bodhisattvas, those considered to be on the path to enlightenment or Nirvana, have given organs and flesh to others to help alleviate grieving and to benefit all. [58] Acceptance varies on applied beliefs and interpretations.
Catholicism does not support embryonic stem cell research, as it entails creation or destruction of human embryos. This destruction conflicts with the belief in the sanctity of life. For example, in the Old Testament, Genesis describes humanity as being created in God’s image and multiplying on the Earth, referencing the sacred rights to human conception and the purpose of development and life. In the Ten Commandments, the tenet that one should not kill has numerous interpretations where killing could mean murder or shedding of the sanctity of life, demonstrating the high value of human personhood. In other books, the theological conception of when life begins is interpreted as in utero, [59] highlighting the inviolability of life and its formation in vivo to make a religious point for accepting such research as relatively limited, if at all. [60] The Vatican has released ethical directives to help apply a theological basis to modern-day conflicts. The Magisterium of the Church states that “unless there is a moral certainty of not causing harm,” experimentation on fetuses, fertilized cells, stem cells, or embryos constitutes a crime. [61] Such procedures would not respect the human person who exists at these stages, according to Catholicism. Damages to the embryo are considered gravely immoral and illicit. [62] Although the Catholic Church officially opposes abortion, surveys demonstrate that many Catholic people hold pro-choice views, whether due to the context of conception, stage of pregnancy, threat to the mother’s life, or for other reasons, demonstrating that practicing members can also accept some but not all tenets. [63]
Some major Jewish denominations, such as the Reform, Conservative, and Reconstructionist movements, are open to supporting ESC use or research as long as it is for saving a life. [64] Within Judaism, the Talmud, or study, gives personhood to the child at birth and emphasizes that life does not begin at conception: [65]
“If she is found pregnant, until the fortieth day it is mere fluid,” [66]
Whereas most religions prioritize the status of human embryos, the Halakah (Jewish religious law) states that to save one life, most other religious laws can be ignored because it is in pursuit of preservation. [67] Stem cell research is accepted due to application of these religious laws.
We recognize that all religions contain subsets and sects. The variety of environmental and cultural differences within religious groups requires further analysis to respect the flexibility of religious thoughts and practices. We make no presumptions that all cultures require notions of autonomy or morality as under the common morality theory , which asserts a set of universal moral norms that all individuals share provides moral reasoning and guides ethical decisions. [68] We only wish to show that the interaction with morality varies between cultures and countries.
III. A Flexible Ethical Approach
The plurality of different moral approaches described above demonstrates that there can be no universally acceptable uniform law for ESC on a global scale. Instead of developing one standard, flexible ethical applications must be continued. We recommend local guidelines that incorporate important cultural and ethical priorities.
While the Declaration of Helsinki is more relevant to people in clinical trials receiving ESC products, in keeping with the tradition of protections for research subjects, consent of the donor is an ethical requirement for ESC donation in many jurisdictions including the US, Canada, and Europe. [69] The Declaration of Helsinki provides a reference point for regulatory standards and could potentially be used as a universal baseline for obtaining consent prior to gamete or embryo donation.
For instance, in Columbia University’s egg donor program for stem cell research, donors followed standard screening protocols and “underwent counseling sessions that included information as to the purpose of oocyte donation for research, what the oocytes would be used for, the risks and benefits of donation, and process of oocyte stimulation” to ensure transparency for consent. [70] The program helped advance stem cell research and provided clear and safe research methods with paid participants. Though paid participation or covering costs of incidental expenses may not be socially acceptable in every culture or context, [71] and creating embryos for ESC research is illegal in many jurisdictions, Columbia’s program was effective because of the clear and honest communications with donors, IRBs, and related stakeholders. This example demonstrates that cultural acceptance of scientific research and of the idea that an egg or embryo does not have personhood is likely behind societal acceptance of donating eggs for ESC research. As noted, many countries do not permit the creation of embryos for research.
Proper communication and education regarding the process and purpose of stem cell research may bolster comprehension and garner more acceptance. “Given the sensitive subject material, a complete consent process can support voluntary participation through trust, understanding, and ethical norms from the cultures and morals participants value. This can be hard for researchers entering countries of different socioeconomic stability, with different languages and different societal values. [72]
An adequate moral foundation in medical ethics is derived from the cultural and religious basis that informs knowledge and actions. [73] Understanding local cultural and religious values and their impact on research could help researchers develop humility and promote inclusion.
IV. Concerns
Some may argue that if researchers all adhere to one ethics standard, protection will be satisfied across all borders, and the global public will trust researchers. However, defining what needs to be protected and how to define such research standards is very specific to the people to which standards are applied. We suggest that applying one uniform guide cannot accurately protect each individual because we all possess our own perceptions and interpretations of social values. [74] Therefore, the issue of not adjusting to the moral pluralism between peoples in applying one standard of ethics can be resolved by building out ethics models that can be adapted to different cultures and religions.
Other concerns include medical tourism, which may promote health inequities. [75] Some countries may develop and approve products derived from ESC research before others, compromising research ethics or drug approval processes. There are also concerns about the sale of unauthorized stem cell treatments, for example, those without FDA approval in the United States. Countries with robust research infrastructures may be tempted to attract medical tourists, and some customers will have false hopes based on aggressive publicity of unproven treatments. [76]
For example, in China, stem cell clinics can market to foreign clients who are not protected under the regulatory regimes. Companies employ a marketing strategy of “ethically friendly” therapies. Specifically, in the case of Beike, China’s leading stem cell tourism company and sprouting network, ethical oversight of administrators or health bureaus at one site has “the unintended consequence of shifting questionable activities to another node in Beike's diffuse network.” [77] In contrast, Jordan is aware of stem cell research’s potential abuse and its own status as a “health-care hub.” Jordan’s expanded regulations include preserving the interests of individuals in clinical trials and banning private companies from ESC research to preserve transparency and the integrity of research practices. [78]
The social priorities of the community are also a concern. The ISSCR explicitly states that guidelines “should be periodically revised to accommodate scientific advances, new challenges, and evolving social priorities.” [79] The adaptable ethics model extends this consideration further by addressing whether research is warranted given the varying degrees of socioeconomic conditions, political stability, and healthcare accessibilities and limitations. An ethical approach would require discussion about resource allocation and appropriate distribution of funds. [80]
While some religions emphasize the sanctity of life from conception, which may lead to public opposition to ESC research, others encourage ESC research due to its potential for healing and alleviating human pain. Many countries have special regulations that balance local views on embryonic personhood, the benefits of research as individual or societal goods, and the protection of human research subjects. To foster understanding and constructive dialogue, global policy frameworks should prioritize the protection of universal human rights, transparency, and informed consent. In addition to these foundational global policies, we recommend tailoring local guidelines to reflect the diverse cultural and religious perspectives of the populations they govern. Ethics models should be adapted to local populations to effectively establish research protections, growth, and possibilities of stem cell research.
For example, in countries with strong beliefs in the moral sanctity of embryos or heavy religious restrictions, an adaptive model can allow for discussion instead of immediate rejection. In countries with limited individual rights and voice in science policy, an adaptive model ensures cultural, moral, and religious views are taken into consideration, thereby building social inclusion. While this ethical consideration by the government may not give a complete voice to every individual, it will help balance policies and maintain the diverse perspectives of those it affects. Embracing an adaptive ethics model of ESC research promotes open-minded dialogue and respect for the importance of human belief and tradition. By actively engaging with cultural and religious values, researchers can better handle disagreements and promote ethical research practices that benefit each society.
This brief exploration of the religious and cultural differences that impact ESC research reveals the nuances of relative ethics and highlights a need for local policymakers to apply a more intense adaptive model.
[1] Poliwoda, S., Noor, N., Downs, E., Schaaf, A., Cantwell, A., Ganti, L., Kaye, A. D., Mosel, L. I., Carroll, C. B., Viswanath, O., & Urits, I. (2022). Stem cells: a comprehensive review of origins and emerging clinical roles in medical practice. Orthopedic reviews , 14 (3), 37498. https://doi.org/10.52965/001c.37498
[2] Poliwoda, S., Noor, N., Downs, E., Schaaf, A., Cantwell, A., Ganti, L., Kaye, A. D., Mosel, L. I., Carroll, C. B., Viswanath, O., & Urits, I. (2022). Stem cells: a comprehensive review of origins and emerging clinical roles in medical practice. Orthopedic reviews , 14 (3), 37498. https://doi.org/10.52965/001c.37498
[3] International Society for Stem Cell Research. (2023). Laboratory-based human embryonic stem cell research, embryo research, and related research activities . International Society for Stem Cell Research. https://www.isscr.org/guidelines/blog-post-title-one-ed2td-6fcdk ; Kimmelman, J., Hyun, I., Benvenisty, N. et al. Policy: Global standards for stem-cell research. Nature 533 , 311–313 (2016). https://doi.org/10.1038/533311a
[4] International Society for Stem Cell Research. (2023). Laboratory-based human embryonic stem cell research, embryo research, and related research activities . International Society for Stem Cell Research. https://www.isscr.org/guidelines/blog-post-title-one-ed2td-6fcdk
[5] Concerning the moral philosophies of stem cell research, our paper does not posit a personal moral stance nor delve into the “when” of human life begins. To read further about the philosophical debate, consider the following sources:
Sandel M. J. (2004). Embryo ethics--the moral logic of stem-cell research. The New England journal of medicine , 351 (3), 207–209. https://doi.org/10.1056/NEJMp048145 ; George, R. P., & Lee, P. (2020, September 26). Acorns and Embryos . The New Atlantis. https://www.thenewatlantis.com/publications/acorns-and-embryos ; Sagan, A., & Singer, P. (2007). The moral status of stem cells. Metaphilosophy , 38 (2/3), 264–284. http://www.jstor.org/stable/24439776 ; McHugh P. R. (2004). Zygote and "clonote"--the ethical use of embryonic stem cells. The New England journal of medicine , 351 (3), 209–211. https://doi.org/10.1056/NEJMp048147 ; Kurjak, A., & Tripalo, A. (2004). The facts and doubts about beginning of the human life and personality. Bosnian journal of basic medical sciences , 4 (1), 5–14. https://doi.org/10.17305/bjbms.2004.3453
[6] Vazin, T., & Freed, W. J. (2010). Human embryonic stem cells: derivation, culture, and differentiation: a review. Restorative neurology and neuroscience , 28 (4), 589–603. https://doi.org/10.3233/RNN-2010-0543
[7] Socially, at its core, the Western approach to ethics is widely principle-based, autonomy being one of the key factors to ensure a fundamental respect for persons within research. For information regarding autonomy in research, see: Department of Health, Education, and Welfare, & National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research (1978). The Belmont Report. Ethical principles and guidelines for the protection of human subjects of research.; For a more in-depth review of autonomy within the US, see: Beauchamp, T. L., & Childress, J. F. (1994). Principles of Biomedical Ethics . Oxford University Press.
[8] Sherley v. Sebelius , 644 F.3d 388 (D.C. Cir. 2011), citing 45 C.F.R. 46.204(b) and [42 U.S.C. § 289g(b)]. https://www.cadc.uscourts.gov/internet/opinions.nsf/6c690438a9b43dd685257a64004ebf99/$file/11-5241-1391178.pdf
[9] Stem Cell Research Enhancement Act of 2005, H. R. 810, 109 th Cong. (2001). https://www.govtrack.us/congress/bills/109/hr810/text ; Bush, G. W. (2006, July 19). Message to the House of Representatives . National Archives and Records Administration. https://georgewbush-whitehouse.archives.gov/news/releases/2006/07/20060719-5.html
[10] National Archives and Records Administration. (2009, March 9). Executive order 13505 -- removing barriers to responsible scientific research involving human stem cells . National Archives and Records Administration. https://obamawhitehouse.archives.gov/the-press-office/removing-barriers-responsible-scientific-research-involving-human-stem-cells
[11] Hurlbut, W. B. (2006). Science, Religion, and the Politics of Stem Cells. Social Research , 73 (3), 819–834. http://www.jstor.org/stable/40971854
[12] Akpa-Inyang, Francis & Chima, Sylvester. (2021). South African traditional values and beliefs regarding informed consent and limitations of the principle of respect for autonomy in African communities: a cross-cultural qualitative study. BMC Medical Ethics . 22. 10.1186/s12910-021-00678-4.
[13] Source for further reading: Tangwa G. B. (2007). Moral status of embryonic stem cells: perspective of an African villager. Bioethics , 21(8), 449–457. https://doi.org/10.1111/j.1467-8519.2007.00582.x , see also Mnisi, F. M. (2020). An African analysis based on ethics of Ubuntu - are human embryonic stem cell patents morally justifiable? African Insight , 49 (4).
[14] Jecker, N. S., & Atuire, C. (2021). Bioethics in Africa: A contextually enlightened analysis of three cases. Developing World Bioethics , 22 (2), 112–122. https://doi.org/10.1111/dewb.12324
[15] Jecker, N. S., & Atuire, C. (2021). Bioethics in Africa: A contextually enlightened analysis of three cases. Developing World Bioethics, 22(2), 112–122. https://doi.org/10.1111/dewb.12324
[16] Jackson, C.S., Pepper, M.S. Opportunities and barriers to establishing a cell therapy programme in South Africa. Stem Cell Res Ther 4 , 54 (2013). https://doi.org/10.1186/scrt204 ; Pew Research Center. (2014, May 1). Public health a major priority in African nations . Pew Research Center’s Global Attitudes Project. https://www.pewresearch.org/global/2014/05/01/public-health-a-major-priority-in-african-nations/
[17] Department of Health Republic of South Africa. (2021). Health Research Priorities (revised) for South Africa 2021-2024 . National Health Research Strategy. https://www.health.gov.za/wp-content/uploads/2022/05/National-Health-Research-Priorities-2021-2024.pdf
[18] Oosthuizen, H. (2013). Legal and Ethical Issues in Stem Cell Research in South Africa. In: Beran, R. (eds) Legal and Forensic Medicine. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-32338-6_80 , see also: Gaobotse G (2018) Stem Cell Research in Africa: Legislation and Challenges. J Regen Med 7:1. doi: 10.4172/2325-9620.1000142
[19] United States Bureau of Citizenship and Immigration Services. (1998). Tunisia: Information on the status of Christian conversions in Tunisia . UNHCR Web Archive. https://webarchive.archive.unhcr.org/20230522142618/https://www.refworld.org/docid/3df0be9a2.html
[20] Gaobotse, G. (2018) Stem Cell Research in Africa: Legislation and Challenges. J Regen Med 7:1. doi: 10.4172/2325-9620.1000142
[21] Kooli, C. Review of assisted reproduction techniques, laws, and regulations in Muslim countries. Middle East Fertil Soc J 24 , 8 (2020). https://doi.org/10.1186/s43043-019-0011-0 ; Gaobotse, G. (2018) Stem Cell Research in Africa: Legislation and Challenges. J Regen Med 7:1. doi: 10.4172/2325-9620.1000142
[22] Pang M. C. (1999). Protective truthfulness: the Chinese way of safeguarding patients in informed treatment decisions. Journal of medical ethics , 25(3), 247–253. https://doi.org/10.1136/jme.25.3.247
[23] Wang, L., Wang, F., & Zhang, W. (2021). Bioethics in China’s biosecurity law: Forms, effects, and unsettled issues. Journal of law and the biosciences , 8(1). https://doi.org/10.1093/jlb/lsab019 https://academic.oup.com/jlb/article/8/1/lsab019/6299199
[24] Wang, Y., Xue, Y., & Guo, H. D. (2022). Intervention effects of traditional Chinese medicine on stem cell therapy of myocardial infarction. Frontiers in pharmacology , 13 , 1013740. https://doi.org/10.3389/fphar.2022.1013740
[25] Li, X.-T., & Zhao, J. (2012). Chapter 4: An Approach to the Nature of Qi in TCM- Qi and Bioenergy. In Recent Advances in Theories and Practice of Chinese Medicine (p. 79). InTech.
[26] Luo, D., Xu, Z., Wang, Z., & Ran, W. (2021). China's Stem Cell Research and Knowledge Levels of Medical Practitioners and Students. Stem cells international , 2021 , 6667743. https://doi.org/10.1155/2021/6667743
[27] Luo, D., Xu, Z., Wang, Z., & Ran, W. (2021). China's Stem Cell Research and Knowledge Levels of Medical Practitioners and Students. Stem cells international , 2021 , 6667743. https://doi.org/10.1155/2021/6667743
[28] Zhang, J. Y. (2017). Lost in translation? accountability and governance of Clinical Stem Cell Research in China. Regenerative Medicine , 12 (6), 647–656. https://doi.org/10.2217/rme-2017-0035
[29] Wang, L., Wang, F., & Zhang, W. (2021). Bioethics in China’s biosecurity law: Forms, effects, and unsettled issues. Journal of law and the biosciences , 8(1). https://doi.org/10.1093/jlb/lsab019 https://academic.oup.com/jlb/article/8/1/lsab019/6299199
[30] Chen, H., Wei, T., Wang, H. et al. Association of China’s two-child policy with changes in number of births and birth defects rate, 2008–2017. BMC Public Health 22 , 434 (2022). https://doi.org/10.1186/s12889-022-12839-0
[31] Azuma, K. Regulatory Landscape of Regenerative Medicine in Japan. Curr Stem Cell Rep 1 , 118–128 (2015). https://doi.org/10.1007/s40778-015-0012-6
[32] Harris, R. (2005, May 19). Researchers Report Advance in Stem Cell Production . NPR. https://www.npr.org/2005/05/19/4658967/researchers-report-advance-in-stem-cell-production
[33] Park, S. (2012). South Korea steps up stem-cell work. Nature . https://doi.org/10.1038/nature.2012.10565
[34] Resnik, D. B., Shamoo, A. E., & Krimsky, S. (2006). Fraudulent human embryonic stem cell research in South Korea: lessons learned. Accountability in research , 13 (1), 101–109. https://doi.org/10.1080/08989620600634193 .
[35] Alahmad, G., Aljohani, S., & Najjar, M. F. (2020). Ethical challenges regarding the use of stem cells: interviews with researchers from Saudi Arabia. BMC medical ethics, 21(1), 35. https://doi.org/10.1186/s12910-020-00482-6
[36] Association for the Advancement of Blood and Biotherapies. https://www.aabb.org/regulatory-and-advocacy/regulatory-affairs/regulatory-for-cellular-therapies/international-competent-authorities/saudi-arabia
[37] Alahmad, G., Aljohani, S., & Najjar, M. F. (2020). Ethical challenges regarding the use of stem cells: Interviews with researchers from Saudi Arabia. BMC medical ethics , 21 (1), 35. https://doi.org/10.1186/s12910-020-00482-6
[38] Alahmad, G., Aljohani, S., & Najjar, M. F. (2020). Ethical challenges regarding the use of stem cells: Interviews with researchers from Saudi Arabia. BMC medical ethics , 21(1), 35. https://doi.org/10.1186/s12910-020-00482-6
Culturally, autonomy practices follow a relational autonomy approach based on a paternalistic deontological health care model. The adherence to strict international research policies and religious pillars within the regulatory environment is a great foundation for research ethics. However, there is a need to develop locally targeted ethics approaches for research (as called for in Alahmad, G., Aljohani, S., & Najjar, M. F. (2020). Ethical challenges regarding the use of stem cells: interviews with researchers from Saudi Arabia. BMC medical ethics, 21(1), 35. https://doi.org/10.1186/s12910-020-00482-6), this decision-making approach may help advise a research decision model. For more on the clinical cultural autonomy approaches, see: Alabdullah, Y. Y., Alzaid, E., Alsaad, S., Alamri, T., Alolayan, S. W., Bah, S., & Aljoudi, A. S. (2022). Autonomy and paternalism in Shared decision‐making in a Saudi Arabian tertiary hospital: A cross‐sectional study. Developing World Bioethics , 23 (3), 260–268. https://doi.org/10.1111/dewb.12355 ; Bukhari, A. A. (2017). Universal Principles of Bioethics and Patient Rights in Saudi Arabia (Doctoral dissertation, Duquesne University). https://dsc.duq.edu/etd/124; Ladha, S., Nakshawani, S. A., Alzaidy, A., & Tarab, B. (2023, October 26). Islam and Bioethics: What We All Need to Know . Columbia University School of Professional Studies. https://sps.columbia.edu/events/islam-and-bioethics-what-we-all-need-know
[39] Ababneh, M. A., Al-Azzam, S. I., Alzoubi, K., Rababa’h, A., & Al Demour, S. (2021). Understanding and attitudes of the Jordanian public about clinical research ethics. Research Ethics , 17 (2), 228-241. https://doi.org/10.1177/1747016120966779
[40] Ababneh, M. A., Al-Azzam, S. I., Alzoubi, K., Rababa’h, A., & Al Demour, S. (2021). Understanding and attitudes of the Jordanian public about clinical research ethics. Research Ethics , 17 (2), 228-241. https://doi.org/10.1177/1747016120966779
[41] Dajani, R. (2014). Jordan’s stem-cell law can guide the Middle East. Nature 510, 189. https://doi.org/10.1038/510189a
[42] Dajani, R. (2014). Jordan’s stem-cell law can guide the Middle East. Nature 510, 189. https://doi.org/10.1038/510189a
[43] The EU’s definition of autonomy relates to the capacity for creating ideas, moral insight, decisions, and actions without constraint, personal responsibility, and informed consent. However, the EU views autonomy as not completely able to protect individuals and depends on other principles, such as dignity, which “expresses the intrinsic worth and fundamental equality of all human beings.” Rendtorff, J.D., Kemp, P. (2019). Four Ethical Principles in European Bioethics and Biolaw: Autonomy, Dignity, Integrity and Vulnerability. In: Valdés, E., Lecaros, J. (eds) Biolaw and Policy in the Twenty-First Century. International Library of Ethics, Law, and the New Medicine, vol 78. Springer, Cham. https://doi.org/10.1007/978-3-030-05903-3_3
[44] Council of Europe. Convention for the protection of Human Rights and Dignity of the Human Being with regard to the Application of Biology and Medicine: Convention on Human Rights and Biomedicine (ETS No. 164) https://www.coe.int/en/web/conventions/full-list?module=treaty-detail&treatynum=164 (forbidding the creation of embryos for research purposes only, and suggests embryos in vitro have protections.); Also see Drabiak-Syed B. K. (2013). New President, New Human Embryonic Stem Cell Research Policy: Comparative International Perspectives and Embryonic Stem Cell Research Laws in France. Biotechnology Law Report , 32 (6), 349–356. https://doi.org/10.1089/blr.2013.9865
[45] Rendtorff, J.D., Kemp, P. (2019). Four Ethical Principles in European Bioethics and Biolaw: Autonomy, Dignity, Integrity and Vulnerability. In: Valdés, E., Lecaros, J. (eds) Biolaw and Policy in the Twenty-First Century. International Library of Ethics, Law, and the New Medicine, vol 78. Springer, Cham. https://doi.org/10.1007/978-3-030-05903-3_3
[46] Tomuschat, C., Currie, D. P., Kommers, D. P., & Kerr, R. (Trans.). (1949, May 23). Basic law for the Federal Republic of Germany. https://www.btg-bestellservice.de/pdf/80201000.pdf
[47] Regulation of Stem Cell Research in Germany . Eurostemcell. (2017, April 26). https://www.eurostemcell.org/regulation-stem-cell-research-germany
[48] Regulation of Stem Cell Research in Finland . Eurostemcell. (2017, April 26). https://www.eurostemcell.org/regulation-stem-cell-research-finland
[49] Regulation of Stem Cell Research in Spain . Eurostemcell. (2017, April 26). https://www.eurostemcell.org/regulation-stem-cell-research-spain
[50] Some sources to consider regarding ethics models or regulatory oversights of other cultures not covered:
Kara MA. Applicability of the principle of respect for autonomy: the perspective of Turkey. J Med Ethics. 2007 Nov;33(11):627-30. doi: 10.1136/jme.2006.017400. PMID: 17971462; PMCID: PMC2598110.
Ugarte, O. N., & Acioly, M. A. (2014). The principle of autonomy in Brazil: one needs to discuss it ... Revista do Colegio Brasileiro de Cirurgioes , 41 (5), 374–377. https://doi.org/10.1590/0100-69912014005013
Bharadwaj, A., & Glasner, P. E. (2012). Local cells, global science: The rise of embryonic stem cell research in India . Routledge.
For further research on specific European countries regarding ethical and regulatory framework, we recommend this database: Regulation of Stem Cell Research in Europe . Eurostemcell. (2017, April 26). https://www.eurostemcell.org/regulation-stem-cell-research-europe
[51] Klitzman, R. (2006). Complications of culture in obtaining informed consent. The American Journal of Bioethics, 6(1), 20–21. https://doi.org/10.1080/15265160500394671 see also: Ekmekci, P. E., & Arda, B. (2017). Interculturalism and Informed Consent: Respecting Cultural Differences without Breaching Human Rights. Cultura (Iasi, Romania) , 14 (2), 159–172.; For why trust is important in research, see also: Gray, B., Hilder, J., Macdonald, L., Tester, R., Dowell, A., & Stubbe, M. (2017). Are research ethics guidelines culturally competent? Research Ethics , 13 (1), 23-41. https://doi.org/10.1177/1747016116650235
[52] The Qur'an (M. Khattab, Trans.). (1965). Al-Mu’minun, 23: 12-14. https://quran.com/23
[53] Lenfest, Y. (2017, December 8). Islam and the beginning of human life . Bill of Health. https://blog.petrieflom.law.harvard.edu/2017/12/08/islam-and-the-beginning-of-human-life/
[54] Aksoy, S. (2005). Making regulations and drawing up legislation in Islamic countries under conditions of uncertainty, with special reference to embryonic stem cell research. Journal of Medical Ethics , 31: 399-403.; see also: Mahmoud, Azza. "Islamic Bioethics: National Regulations and Guidelines of Human Stem Cell Research in the Muslim World." Master's thesis, Chapman University, 2022. https://doi.org/10.36837/ chapman.000386
[55] Rashid, R. (2022). When does Ensoulment occur in the Human Foetus. Journal of the British Islamic Medical Association , 12 (4). ISSN 2634 8071. https://www.jbima.com/wp-content/uploads/2023/01/2-Ethics-3_-Ensoulment_Rafaqat.pdf.
[56] Sivaraman, M. & Noor, S. (2017). Ethics of embryonic stem cell research according to Buddhist, Hindu, Catholic, and Islamic religions: perspective from Malaysia. Asian Biomedicine,8(1) 43-52. https://doi.org/10.5372/1905-7415.0801.260
[57] Jafari, M., Elahi, F., Ozyurt, S. & Wrigley, T. (2007). 4. Religious Perspectives on Embryonic Stem Cell Research. In K. Monroe, R. Miller & J. Tobis (Ed.), Fundamentals of the Stem Cell Debate: The Scientific, Religious, Ethical, and Political Issues (pp. 79-94). Berkeley: University of California Press. https://escholarship.org/content/qt9rj0k7s3/qt9rj0k7s3_noSplash_f9aca2e02c3777c7fb76ea768ba458f0.pdf https://doi.org/10.1525/9780520940994-005
[58] Lecso, P. A. (1991). The Bodhisattva Ideal and Organ Transplantation. Journal of Religion and Health , 30 (1), 35–41. http://www.jstor.org/stable/27510629 ; Bodhisattva, S. (n.d.). The Key of Becoming a Bodhisattva . A Guide to the Bodhisattva Way of Life. http://www.buddhism.org/Sutras/2/BodhisattvaWay.htm
[59] There is no explicit religious reference to when life begins or how to conduct research that interacts with the concept of life. However, these are relevant verses pertaining to how the fetus is viewed. (( King James Bible . (1999). Oxford University Press. (original work published 1769))
Jerimiah 1: 5 “Before I formed thee in the belly I knew thee; and before thou camest forth out of the womb I sanctified thee…”
In prophet Jerimiah’s insight, God set him apart as a person known before childbirth, a theme carried within the Psalm of David.
Psalm 139: 13-14 “…Thou hast covered me in my mother's womb. I will praise thee; for I am fearfully and wonderfully made…”
These verses demonstrate David’s respect for God as an entity that would know of all man’s thoughts and doings even before birth.
[60] It should be noted that abortion is not supported as well.
[61] The Vatican. (1987, February 22). Instruction on Respect for Human Life in Its Origin and on the Dignity of Procreation Replies to Certain Questions of the Day . Congregation For the Doctrine of the Faith. https://www.vatican.va/roman_curia/congregations/cfaith/documents/rc_con_cfaith_doc_19870222_respect-for-human-life_en.html
[62] The Vatican. (2000, August 25). Declaration On the Production and the Scientific and Therapeutic Use of Human Embryonic Stem Cells . Pontifical Academy for Life. https://www.vatican.va/roman_curia/pontifical_academies/acdlife/documents/rc_pa_acdlife_doc_20000824_cellule-staminali_en.html ; Ohara, N. (2003). Ethical Consideration of Experimentation Using Living Human Embryos: The Catholic Church’s Position on Human Embryonic Stem Cell Research and Human Cloning. Department of Obstetrics and Gynecology . Retrieved from https://article.imrpress.com/journal/CEOG/30/2-3/pii/2003018/77-81.pdf.
[63] Smith, G. A. (2022, May 23). Like Americans overall, Catholics vary in their abortion views, with regular mass attenders most opposed . Pew Research Center. https://www.pewresearch.org/short-reads/2022/05/23/like-americans-overall-catholics-vary-in-their-abortion-views-with-regular-mass-attenders-most-opposed/
[64] Rosner, F., & Reichman, E. (2002). Embryonic stem cell research in Jewish law. Journal of halacha and contemporary society , (43), 49–68.; Jafari, M., Elahi, F., Ozyurt, S. & Wrigley, T. (2007). 4. Religious Perspectives on Embryonic Stem Cell Research. In K. Monroe, R. Miller & J. Tobis (Ed.), Fundamentals of the Stem Cell Debate: The Scientific, Religious, Ethical, and Political Issues (pp. 79-94). Berkeley: University of California Press. https://escholarship.org/content/qt9rj0k7s3/qt9rj0k7s3_noSplash_f9aca2e02c3777c7fb76ea768ba458f0.pdf https://doi.org/10.1525/9780520940994-005
[65] Schenker J. G. (2008). The beginning of human life: status of embryo. Perspectives in Halakha (Jewish Religious Law). Journal of assisted reproduction and genetics , 25 (6), 271–276. https://doi.org/10.1007/s10815-008-9221-6
[66] Ruttenberg, D. (2020, May 5). The Torah of Abortion Justice (annotated source sheet) . Sefaria. https://www.sefaria.org/sheets/234926.7?lang=bi&with=all&lang2=en
[67] Jafari, M., Elahi, F., Ozyurt, S. & Wrigley, T. (2007). 4. Religious Perspectives on Embryonic Stem Cell Research. In K. Monroe, R. Miller & J. Tobis (Ed.), Fundamentals of the Stem Cell Debate: The Scientific, Religious, Ethical, and Political Issues (pp. 79-94). Berkeley: University of California Press. https://escholarship.org/content/qt9rj0k7s3/qt9rj0k7s3_noSplash_f9aca2e02c3777c7fb76ea768ba458f0.pdf https://doi.org/10.1525/9780520940994-005
[68] Gert, B. (2007). Common morality: Deciding what to do . Oxford Univ. Press.
[69] World Medical Association (2013). World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA , 310(20), 2191–2194. https://doi.org/10.1001/jama.2013.281053 Declaration of Helsinki – WMA – The World Medical Association .; see also: National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. (1979). The Belmont report: Ethical principles and guidelines for the protection of human subjects of research . U.S. Department of Health and Human Services. https://www.hhs.gov/ohrp/regulations-and-policy/belmont-report/read-the-belmont-report/index.html
[70] Zakarin Safier, L., Gumer, A., Kline, M., Egli, D., & Sauer, M. V. (2018). Compensating human subjects providing oocytes for stem cell research: 9-year experience and outcomes. Journal of assisted reproduction and genetics , 35 (7), 1219–1225. https://doi.org/10.1007/s10815-018-1171-z https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6063839/ see also: Riordan, N. H., & Paz Rodríguez, J. (2021). Addressing concerns regarding associated costs, transparency, and integrity of research in recent stem cell trial. Stem Cells Translational Medicine , 10 (12), 1715–1716. https://doi.org/10.1002/sctm.21-0234
[71] Klitzman, R., & Sauer, M. V. (2009). Payment of egg donors in stem cell research in the USA. Reproductive biomedicine online , 18 (5), 603–608. https://doi.org/10.1016/s1472-6483(10)60002-8
[72] Krosin, M. T., Klitzman, R., Levin, B., Cheng, J., & Ranney, M. L. (2006). Problems in comprehension of informed consent in rural and peri-urban Mali, West Africa. Clinical trials (London, England) , 3 (3), 306–313. https://doi.org/10.1191/1740774506cn150oa
[73] Veatch, Robert M. Hippocratic, Religious, and Secular Medical Ethics: The Points of Conflict . Georgetown University Press, 2012.
[74] Msoroka, M. S., & Amundsen, D. (2018). One size fits not quite all: Universal research ethics with diversity. Research Ethics , 14 (3), 1-17. https://doi.org/10.1177/1747016117739939
[75] Pirzada, N. (2022). The Expansion of Turkey’s Medical Tourism Industry. Voices in Bioethics , 8 . https://doi.org/10.52214/vib.v8i.9894
[76] Stem Cell Tourism: False Hope for Real Money . Harvard Stem Cell Institute (HSCI). (2023). https://hsci.harvard.edu/stem-cell-tourism , See also: Bissassar, M. (2017). Transnational Stem Cell Tourism: An ethical analysis. Voices in Bioethics , 3 . https://doi.org/10.7916/vib.v3i.6027
[77] Song, P. (2011) The proliferation of stem cell therapies in post-Mao China: problematizing ethical regulation, New Genetics and Society , 30:2, 141-153, DOI: 10.1080/14636778.2011.574375
[78] Dajani, R. (2014). Jordan’s stem-cell law can guide the Middle East. Nature 510, 189. https://doi.org/10.1038/510189a
[79] International Society for Stem Cell Research. (2024). Standards in stem cell research . International Society for Stem Cell Research. https://www.isscr.org/guidelines/5-standards-in-stem-cell-research
[80] Benjamin, R. (2013). People’s science bodies and rights on the Stem Cell Frontier . Stanford University Press.
Mifrah Hayath
SM Candidate Harvard Medical School, MS Biotechnology Johns Hopkins University
Olivia Bowers
MS Bioethics Columbia University (Disclosure: affiliated with Voices in Bioethics)
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License .
Suggestions or feedback?
MIT News | Massachusetts Institute of Technology
- Machine learning
- Social justice
- Black holes
- Classes and programs
Departments
- Aeronautics and Astronautics
- Brain and Cognitive Sciences
- Architecture
- Political Science
- Mechanical Engineering
Centers, Labs, & Programs
- Abdul Latif Jameel Poverty Action Lab (J-PAL)
- Picower Institute for Learning and Memory
- Lincoln Laboratory
- School of Architecture + Planning
- School of Engineering
- School of Humanities, Arts, and Social Sciences
- Sloan School of Management
- School of Science
- MIT Schwarzman College of Computing
New treatment could reverse hair loss caused by an autoimmune skin disease
Press contact :, media download.

*Terms of Use:
Images for download on the MIT News office website are made available to non-commercial entities, press and the general public under a Creative Commons Attribution Non-Commercial No Derivatives license . You may not alter the images provided, other than to crop them to size. A credit line must be used when reproducing images; if one is not provided below, credit the images to "MIT."

Previous image Next image
Researchers at MIT, Brigham and Women’s Hospital, and Harvard Medical School have developed a potential new treatment for alopecia areata, an autoimmune disorder that causes hair loss and affects people of all ages, including children.
For most patients with this type of hair loss, there is no effective treatment. The team developed a microneedle patch that can be painlessly applied to the scalp and releases drugs that help to rebalance the immune response at the site, halting the autoimmune attack.
In a study of mice, the researchers found that this treatment allowed hair to regrow and dramatically reduced inflammation at the treatment site, while avoiding systemic immune effects elsewhere in the body. This strategy could also be adapted to treat other autoimmune skin diseases such as vitiligo, atopic dermatitis, and psoriasis, the researchers say.
“This innovative approach marks a paradigm shift. Rather than suppressing the immune system, we’re now focusing on regulating it precisely at the site of antigen encounter to generate immune tolerance,” says Natalie Artzi, a principal research scientist in MIT’s Institute for Medical Engineering and Science, an associate professor of medicine at Harvard Medical School and Brigham and Women’s Hospital, and an associate faculty member at the Wyss Institute of Harvard University.
Artzi and Jamil R. Azzi, an associate professor of medicine at Harvard Medical School and Brigham and Women’s Hospital, are the senior authors of the new study , which appears in the journal Advanced Materials . Nour Younis, a Brigham and Women’s postdoc, and Nuria Puigmal, a Brigham and Women’s postdoc and former MIT research affiliate, are the lead authors of the paper.
The researchers are now working on launching a company to further develop the technology, led by Puigmal, who was recently awarded a Harvard Business School Blavatnik Fellowship.
Direct delivery
Alopecia areata, which affects more than 6 million Americans, occurs when the body’s own T cells attack hair follicles, leading the hair to fall out. The only treatment available to most patients — injections of immunosuppressant steroids into the scalp — is painful and patients often can’t tolerate it.
Some patients with alopecia areata and other autoimmune skin diseases can also be treated with immunosuppressant drugs that are given orally, but these drugs lead to widespread suppression of the immune system, which can have adverse side effects.
“This approach silences the entire immune system, offering relief from inflammation symptoms but leading to frequent recurrences. Moreover, it increases susceptibility to infections, cardiovascular diseases, and cancer,” Artzi says.
A few years ago, at a working group meeting in Washington, Artzi happened to be seated next to Azzi (the seating was alphabetical), an immunologist and transplant physican who was seeking new ways to deliver drugs directly to the skin to treat skin-related diseases.
Their conversation led to a new collaboration, and the two labs joined forces to work on a microneedle patch to deliver drugs to the skin. In 2021, they reported that such a patch can be used to prevent rejection following skin transplant. In the new study, they began applying this approach to autoimmune skin disorders.
“The skin is the only organ in our body that we can see and touch, and yet when it comes to drug delivery to the skin, we revert to systemic administration. We saw great potential in utilizing the microneedle patch to reprogram the immune system locally,” Azzi says.
The microneedle patches used in this study are made from hyaluronic acid crosslinked with polyethylene glycol (PEG), both of which are biocompatible and commonly used in medical applications. With this delivery method, drugs can pass through the tough outer layer of the epidermis, which can’t be penetrated by creams applied to the skin.
“This polymer formulation allows us to create highly durable needles capable of effectively penetrating the skin. Additionally, it gives us the flexibility to incorporate any desired drug,” Artzi says. For this study, the researchers loaded the patches with a combination of the cytokines IL-2 and CCL-22. Together, these immune molecules help to recruit regulatory T cells, which proliferate and help to tamp down inflammation. These cells also help the immune system learn to recognize that hair follicles are not foreign antigens, so that it will stop attacking them.
Hair regrowth
The researchers found that mice treated with this patch every other day for three weeks had many more regulatory T cells present at the site, along with a reduction in inflammation. Hair was able to regrow at those sites, and this growth was maintained for several weeks after the treatment ended. In these mice, there were no changes in the levels of regulatory T cells in the spleen or lymph nodes, suggesting that the treatment affected only the site where the patch was applied.
In another set of experiments, the researchers grafted human skin onto mice with a humanized immune system. In these mice, the microneedle treatment also induced proliferation of regulatory T cells and a reduction in inflammation.
The researchers designed the microneedle patches so that after releasing their drug payload, they can also collect samples that could be used to monitor the progress of the treatment. Hyaluronic acid causes the needles to swell about tenfold after entering the skin, which allows them to absorb interstitial fluid containing biomolecules and immune cells from the skin.
Following patch removal, researchers can analyze samples to measure levels of regulatory T cells and inflammation markers. This could prove valuable for monitoring future patients who may undergo this treatment.
The researchers now plan to further develop this approach for treating alopecia, and to expand into other autoimmune skin diseases.
The research was funded by the Ignite Fund and Shark Tank Fund awards from the Department of Medicine at Brigham and Women’s Hospital.
Share this news article on:
Press mentions, healthday news.
MIT researchers have developed microneedle patches that are capable of restoring hair growth in alopecia areata patients, reports Ernie Mundell for HealthDay . The team’s approach includes a, “patch containing myriad microneedles that is applied to the scalp,” writes Mundell. “It releases drugs to reset the immune system so it stops attacking follicles.”
Previous item Next item
Related Links
- Natalie Artzi
- Institute for Medical Engineering and Science
Related Topics
- Drug delivery
- Health sciences and technology
- Institute for Medical Engineering and Science (IMES)
Related Articles

A sprayable gel could make minimally invasive surgeries simpler and safer
Patch that delivers drug, gene, and light-based therapy to tumor sites shows promising results

MIT researchers design tailored tissue adhesives
More mit news.
Janabel Xia: Algorithms, dance rhythms, and the drive to succeed
Read full story →

Jonathan Byrnes, MIT Center for Transportation and Logistics senior lecturer and visionary in supply chain management, dies at 75

Researchers develop a detector for continuously monitoring toxic gases

The beauty of biology

Navigating longevity with industry leaders at MIT AgeLab PLAN Forum

Jeong Min Park earns 2024 Schmidt Science Fellowship
- More news on MIT News homepage →
Massachusetts Institute of Technology 77 Massachusetts Avenue, Cambridge, MA, USA
- Map (opens in new window)
- Events (opens in new window)
- People (opens in new window)
- Careers (opens in new window)
- Accessibility
- Social Media Hub
- MIT on Facebook
- MIT on YouTube
- MIT on Instagram

IMAGES
VIDEO
COMMENTS
Abstract. Stem cell therapies are being explored extensively as treatments for degenerative eye disease, either for replacing lost neurons, restoring neural circuits or, based on more recent evidence, as paracrine-mediated therapies in which stem cell-derived trophic factors protect compromised endogenous retinal neurons from death and induce ...
Stem cell therapy in ocular diseases certainly represents the future in the treatment of numerous serious eye diseases, especially if organ transplantation can be avoided. One of the main problems of these therapies, mainly the immunological tolerance to transplanted cells, has been solved thanks to the use of autologous cells.
These latest stem cell transplantation studies showed that this therapy is a promising approach to restore visual function in eyes with degenerative retinal diseases such as retinitis pigmentosa, Stargardts' macular dystrophy, and age-related macular degeneration. This review focuses on new developments in stem cell therapy for degenerative ...
Debilitating diseases of the eye represent a large unmet medical need potentially addressable with stem cell-based approaches. Over the past decade, the California Institute for Regenerative Medicine (CIRM) has funded and supported the translation, from early research concepts to human trials, of therapeutic stem cell approaches for dry age-related macular degeneration, retinitis pigmentosa ...
Finally, the eye's transparent optics make it possible to image the retina at the single-cell level in animals and humans, enabling monitoring of disease and treatment responses.
Jurkunas's study is one of several taking place worldwide that use stem cells to repair damaged corneas. Other researchers are trying to regenerate cells in the retina — the main light-sensing ...
Herein, we review the safety, efficacy, regulatory standards and ethical implications of the use of stem cells in ocular disease. A literature review was conducted, registered clinical trials reviewed, and expert opinions sought. Guidelines and codes of conduct from international societies and profe …
There are currently many clinical trials ongoing which aim to test the safety and efficacy of stem cell transplantation in the eye (Table 1).This review focuses on the potential of non retinal-derived stem cells, in particular NSC, BMSC, ADSC, DPSC and ESC/iPSC for the treatment of traumatic and degenerative eye disease and, where relevant, inter-relates some findings from stem cell research ...
Abstract. Recent advancements in stem cell technology open a new door for patients suffering from diseases and disorders that have yet to be treated. Stem cell-based therapy, including human ...
Abstract. There is a rise in the number of people who have vision loss due to retinal diseases, and conventional therapies for treating retinal degeneration fail to repair and regenerate the damaged retina. Several studies in animal models and human trials have explored the use of stem cells to repair the retinal tissue to improve visual acuity.
Stem Cells: New Opportunities to Treat Eye Diseases. Iqbal Ahmad. Author Affiliations. Investigative Ophthalmology & Visual Science November 2001, Vol.42, 2743-2748. doi: The recent identification and characterization of neural progenitors with stem cell properties has opened new avenues that may be useful for treating functional impairments ...
The nature of our research in stem cell differentiation, treatment of retinal degenerative diseases, and usage of gene editing technologies to create disease models (Deng et al., 2018, Jin et al., 2012, Lin et al., 2020, Liu et al., 2020, Lv et al., 2017, Pan et al., 2021, Zhang et al., 2021b), we paid particular attention to the advancement in ...
Purpose of Review In this article, we review recent studies that examine stem cells as a potential treatment for ocular surface diseases. Recent Findings Mesenchymal stem cells (MSCs) derived from non-ocular surface tissues were effective in treating limbal stem cell deficiency and corneal endothelial dysfunction. In dry eye, limbal stem cells reduced ocular symptoms, while MSCs improved tear ...
Investigators from the Board of Governors Regenerative Medicine Institute at Cedars-Sinai are advancing stem cell technology to treat degenerative diseases of the eye. In one recent study, they determined the optimal dose and surgical method for transplanting cells into the subretinal space, providing the basis for an ongoing clinical trial in patients with retinitis pigmentosa.
Bharti is leading the development of patient stem cell-derived RPE for an AMD clinical trial set to launch in 2018. In a healthy eye, RPE cells nourish and support photoreceptors, the cells that convert light into electrical signals that travel to the brain via the optic nerve. RPE cells form a layer just behind the photoreceptors.
Stem cells of eye are closely associated with maxillofacial tissues including dental stem cells (a derivative of CNCC) during embryogenesis which retain the stem cells till life can be traced and reprogrammed for stem cell therapy. Researchers differentiated retina [ 68] from dental pulp stem cells.
Researchers at the Jules Stein Eye Institute (JSEI) have begun two clinical trials to determine the safety of stem cell therapy and patients' ability to tolerate it for the treatment of two common, currently untreatable degenerative eye diseases. This FDA-approved study is only the second in the United States to use human embryonic-derived stem cells in patients and the first to address eye ...
Role of stem cells in regenerative treatment of dry eye disease caused by lacrimal gland dysfunction. ... and conjunctival epithelial cells. Dry eye disease (DED), also known as keratoconjunctivitis sicca, is a chronic multifactorial disease, ... there have been two main areas of research aimed at regeneration of the LG: (1) ...
Dec. 22, 2022 — Scientists used patient stem cells and 3D bioprinting to produce eye tissue that will advance understanding of the mechanisms of blinding diseases. The research team printed a ...
A decade ago, clinicians had nothing to offer most people affected by retinal degeneration. Breakthroughs in genetics, bionics and stem-cell therapy are changing that. Worldwide, 36 million people ...
New IRP research could make it much easier to grow organoids that mimic the eye's retina, thereby accelerating discoveries about a variety of vision-impairing diseases. 1 Retinal organoids are three-dimensional structures with distinct layers containing all the major cell types found in the real human retina, the part of the eye that converts ...
Credit: Science Photo Library. A CRISPR-based gene-editing therapy led to improved vision in people with an inherited condition that causes blindness 1. Mutations in more than 20 genes can lead to ...
Stem cell therapies are getting headlines for their potential to cure diseases, including those that affect vision. But an important message is missing: the therapies are not yet proven to be safe and effective for your eyes. Stem cell treatments appear to offer hope to people with few options to recover vision.
The use of stem cell technology to reverse blindness promises great individual and societal benefit. Stem cell therapies seek to replace or enhance the function of damaged tissues. Currently, there are no FDA approved and commercially available products in this arena. Numerous clinical trials are underway, but as of today, we are still awaiting ...
The trial, which started in 2019, enrolled 12 adults, ages 17 to 63, and two children, ages 9 and 14, with inherited retinal degeneration caused by mutations in the CEP290 gene. That gene provides ...
Stem cells, characterized by their unique ability to differentiate into various cell types, enable the repair or replacement of damaged tissues. Two primary types of stem cells are somatic stem cells (adult stem cells) and embryonic stem cells. Adult stem cells exist in developed tissues and maintain the body's repair processes. [1]
The researchers are now working on launching a company to further develop the technology, led by Puigmal, who was recently awarded a Harvard Business School Blavatnik Fellowship. Direct delivery. Alopecia areata, which affects more than 6 million Americans, occurs when the body's own T cells attack hair follicles, leading the hair to fall out.
CAR T-cell immunotherapy changes these collected T cells to produce chimeric antigen receptors (or CARs) that adhere to tumors to destroy them. A recent study published in the New England Journal of Medicine, suggests CAR T-cell therapy could become a highly effective treatment for SLE patients who do not respond to current lupus therapeutics ...